


1 / 5










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |


| Model Number | Spec.(mm) |
|---|---|
| 1072001 | S |
| M | |
| L |

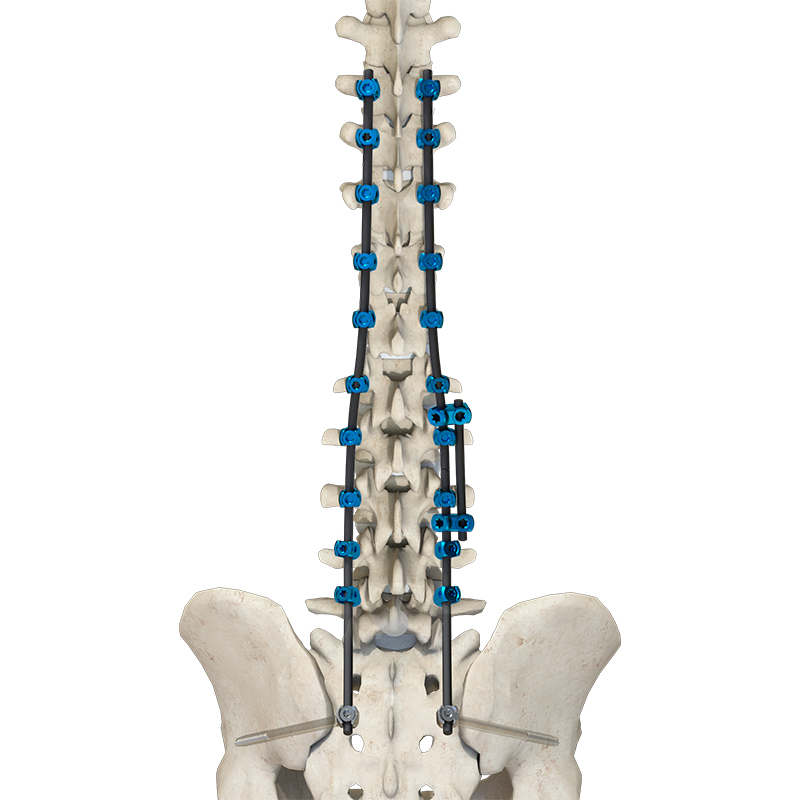
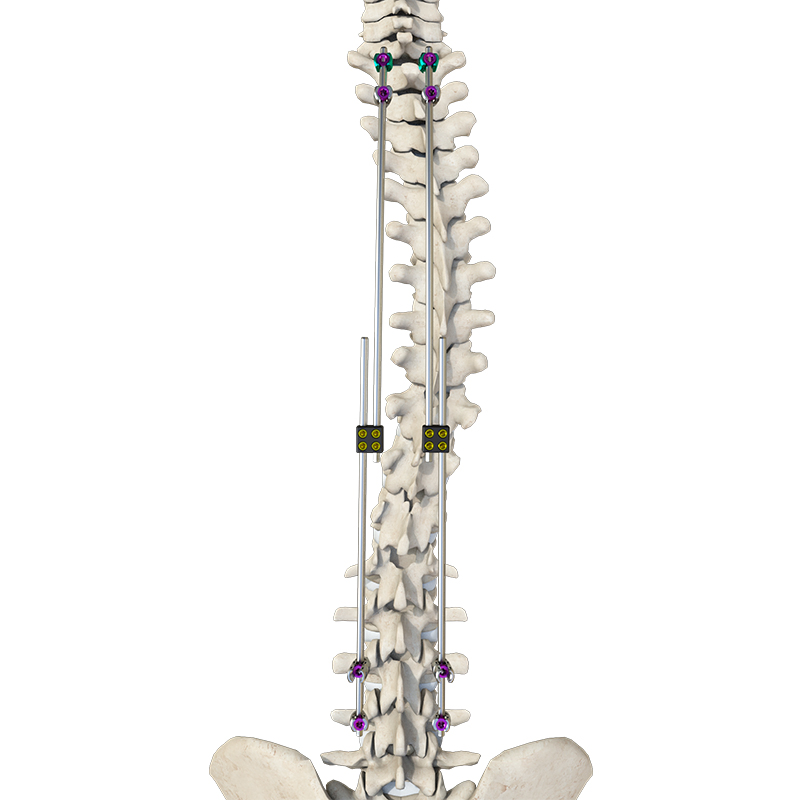
 Daher Orthopedic Implants
Daher Orthopedic Implants