The global market for intramedullary (IM) nails is witnessing unprecedented growth, driven by an aging global population and the increasing incidence of trauma-related injuries. As the "gold standard" for the treatment of long bone fractures, such as those in the femur, tibia, and humerus, intramedullary nails offer superior biomechanical advantages including load-sharing and minimally invasive insertion.
Today, the orthopedic community prioritizes implants that promote early mobilization and rapid healing. The industry is valued at billions of dollars, with North America and Europe currently leading in consumption, while the Asia-Pacific region is emerging as the fastest-growing hub for production and surgical adoption. We, as a specialized manufacturer, sit at the heart of this supply chain, delivering high-grade titanium and stainless steel solutions to healthcare providers worldwide.
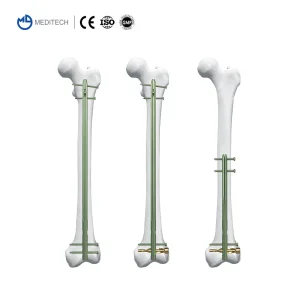
Modern IM nails are designed for suprapatellar and retrograde approaches, reducing soft tissue damage and post-operative pain for patients.
Advanced multi-locking options and "expert" nail designs allow for better stability in distal and proximal metaphyseal fractures.
Transitioning from standard stainless steel to Grade 5 Titanium alloys (Ti6Al4V) to ensure better biocompatibility and reduced MRI interference.
Our Suprapatellar approach system represents the pinnacle of tibial fracture fixation. By allowing the surgeon to work with the leg in a semi-extended position, it simplifies the use of fluoroscopy and reduces the strain on the patellar tendon. Manufactured from high-strength titanium, this nail offers various interlocking options to stabilize complex fractures.
Medical distributors and hospital procurement departments require more than just a product; they require a reliable supply chain partner. Global demand centers on consistency, regulatory compliance (CE/FDA), and cost-effectiveness.
We understand the complexities of international tenders. Our production facility is equipped with high-precision CNC machines that ensure every batch of PFNA (Proximal Femoral Nail Antirotation) or Humeral nails meets exacting tolerances. By maintaining large inventories and optimized logistics, we ensure that trauma centers receive their supplies without delay, even in high-demand periods.

Leveraging China's mature industrial ecosystem, we provide premium orthopedic implants at a fraction of the cost of Western brands without compromising on material quality.
Our in-house design team works directly with clinical experts to refine nail geometries and locking mechanisms based on real-world surgical feedback.
From small-batch custom implants to massive volume orders for national healthcare systems, our factory is built to scale quickly.
The PFNA (Proximal Femoral Nail Antirotation) is a cornerstone of geriatric trauma care. Our system features a helical blade that achieves superior compaction of the cancellous bone, providing significantly higher resistance to cut-out—a critical factor in osteoporotic patients. Available in various lengths and diameters to suit diverse patient morphologies.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential. Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications.
For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia. Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.

The clinical application of intramedullary nails has revolutionized the way orthopedic surgeons manage diaphyseal fractures. Unlike traditional plate fixation, which requires extensive surgical exposure and disrupts the periosteal blood supply, intramedullary nailing is a centromedullary, load-sharing technique. This means the nail shares the mechanical stress with the bone, allowing for controlled micromotion at the fracture site, which is known to stimulate callus formation and accelerate secondary bone healing.
In the global commercial landscape, the shift towards Titanium alloys has been significant. Titanium's modulus of elasticity is closer to that of human bone compared to stainless steel, which reduces the "stress shielding" effect. Furthermore, the development of specialized coatings and surface treatments has improved the biocompatibility and long-term stability of these implants. As a leading exporter, we ensure our titanium nails undergo rigorous fatigue testing to simulate the repetitive loads of walking and limb movement.
Localized application scenarios often dictate the specific design requirements of the nails. For instance, in regions with higher rates of high-energy trauma (such as motor vehicle accidents), there is a greater demand for interlocking femoral nails with multiple proximal and distal locking options to address comminuted fractures. In contrast, in regions with an aging population, the focus shifts toward PFNA (Proximal Femoral Nail Antirotation) systems that are optimized for osteoporotic bone, where traditional screws might lack sufficient purchase.
Our role as a China-based factory allows us to bridge the gap between high-tech medical engineering and economic feasibility. We utilize 5-axis CNC machining centers to produce complex geometries, such as the anatomic bow of the femoral nail, which is crucial for preventing cortical perforation during insertion. Our commitment to the "Expert" series of nails—featuring end caps to prevent tissue ingrowth and cannulated designs for guide wire compatibility—ensures that surgeons have the tools they need for a seamless procedure.
The future of intramedullary nailing lies in "Smart Implants." We are currently exploring the integration of biodegradable materials and antimicrobial coatings to further reduce the risk of post-operative infections—one of the most challenging complications in trauma surgery. By choosing a manufacturer with a robust R&D foundation, you are investing in the future of patient care.
Whether it is the Suprapatellar Tibial Nail for improved knee joint preservation or the Multi-lock Humeral Nail for complex upper limb fractures, our portfolio is designed to meet the rigorous demands of modern surgery. We invite global partners, from hospital groups to medical device distributors, to explore our manufacturing capabilities and join us in our mission to restore mobility to patients around the world.