




1 / 5










| Still deciding? Get samples of $ ! US$ 24/Piece Request Sample |
| Product Number | Product Name | Material | Size (mm) |
|---|---|---|---|
| 73601 | KCO Laminar Hook 736 | Titanium Alloy | 4.5mm |
| 73602 | 6mm |

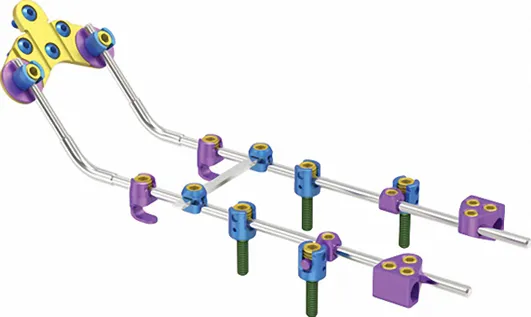

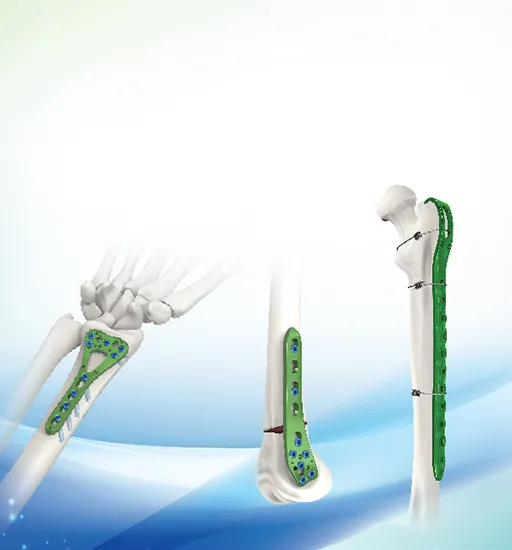


We are a leading manufacturer of orthopedic implants and instruments since 1958. Our main product range includes trauma plates, interlocking nails, spinal fixators, external fixators, and power surgical tools. All products are manufactured under strict quality control systems and adhere to international standards.

 Daher Orthopedic Implants
Daher Orthopedic Implants