













| Still deciding? Get samples of $ ! US$ 20.3/Piece Request Sample |

Available Sizes:
| Product Name | Double Head Hook |
| Reference Code | 686 |
Related Products:
Certifications:



Exhibition Gallery:



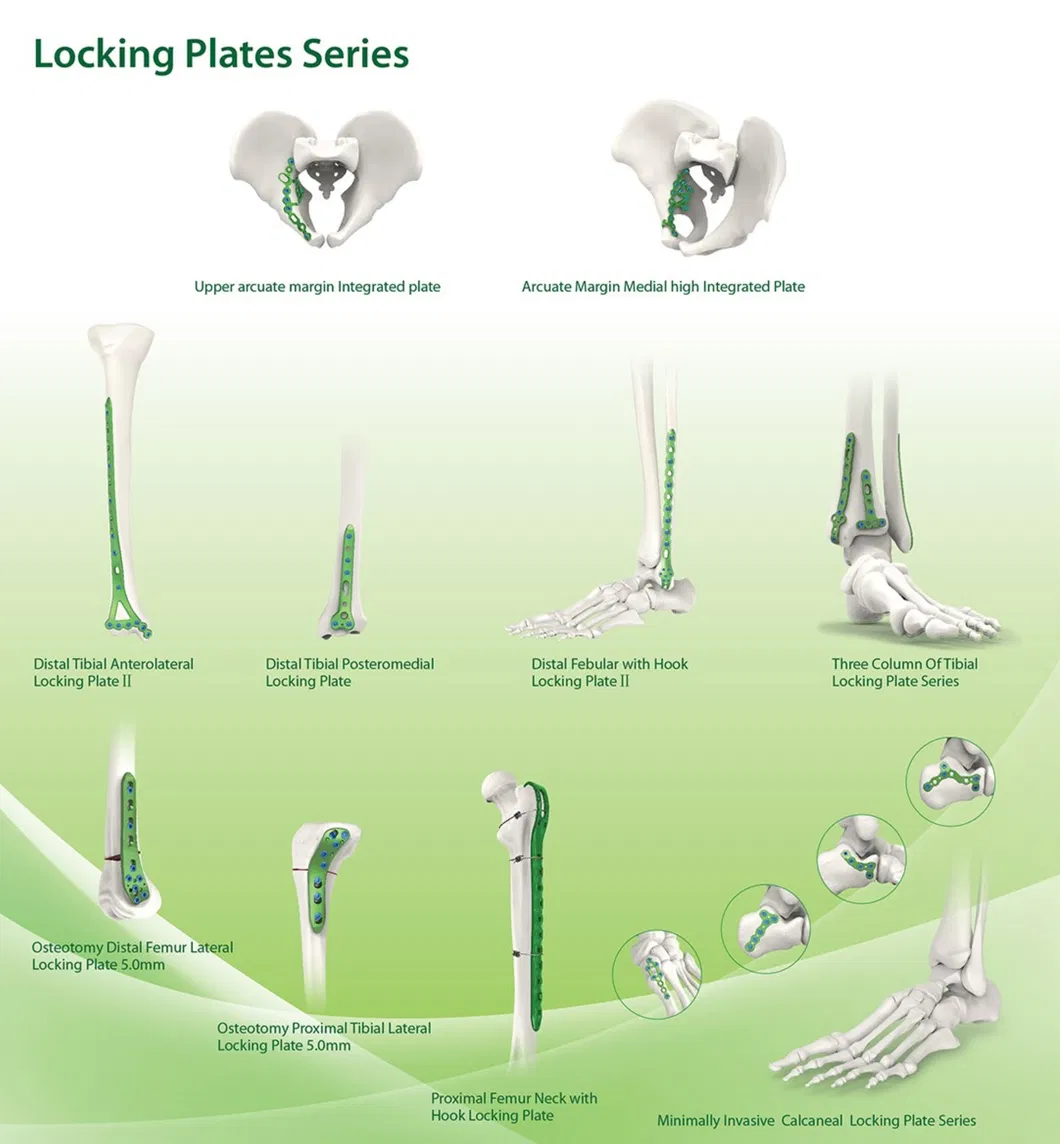
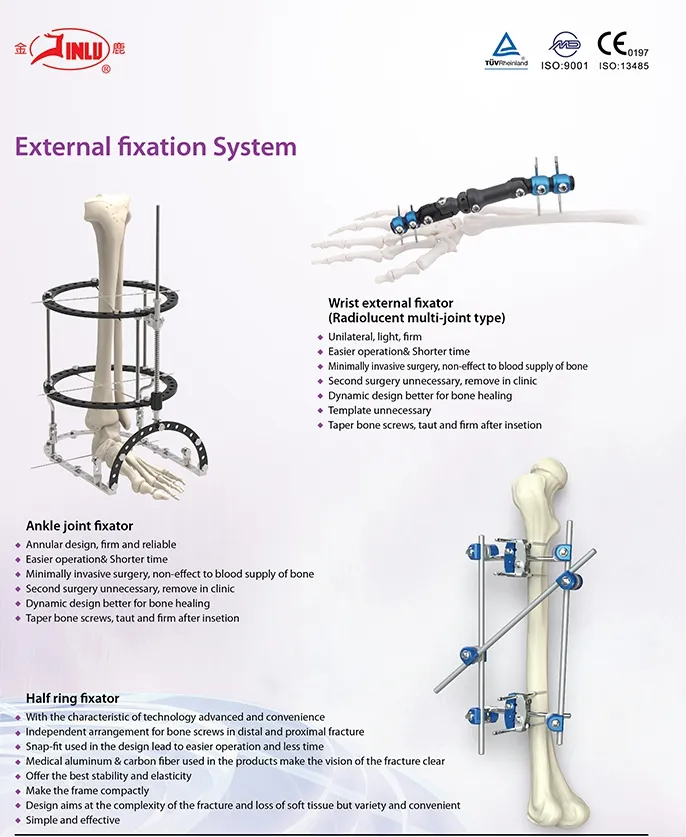
Established in 1958, our company is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. We are recognized as a key enterprise for medical apparatus, specializing in a wide range of products including Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis, and specialized surgical instruments.
Our products are known for their multi-varieties and complete specifications. All medical apparatus have passed rigorous national and provincial authentications. Each product is marked with a material code and anti-fake laser marks, ensuring traceability and quality responsibility.
Equipped with advanced imported CNC machines and high-precision digital control systems, we maintain a 100,000 Grade axenic purification chamber to ensure the highest quality standards. We are ISO 9001 certified, and our products have received numerous national silver awards and science and technology honors.
We adhere to the tenet of "creating high quality with science and technology." We strive for perfect design, innovative technology, and top-ranking products combined with comprehensive service for all our global partners.

 Daher Orthopedic Implants
Daher Orthopedic Implants