In the contemporary medical industry, Titanium Bone Screws have emerged as the gold standard for fracture fixation and reconstructive surgery. The global market for orthopedic implants is witnessing a significant paradigm shift, driven by an aging population and an increase in sports-related injuries. Titanium, specifically Grade 5 (Ti-6Al-4V) and pure titanium, is favored for its extraordinary biocompatibility, high strength-to-weight ratio, and excellent corrosion resistance within the human body.
As a leading Titanium Bone Screws supplier and exporter, we observe that the demand is no longer just about basic fixation. Modern surgeons require precision-engineered threads, varying pitch designs for optimal compression, and surface treatments that promote osseointegration. From the United States to Germany, and across the emerging healthcare hubs in Asia, the shift toward minimally invasive surgery (MIS) has necessitated the development of specialized screws like the headless compression and cannulated varieties.

Recent developments focus on bioactive coatings such as Hydroxyapatite (HA) and Antimicrobial coatings. These technologies aim to speed up the bone-healing process and reduce the risk of post-operative infections, a critical factor for global procurement teams.
The transition from traditional CNC machining to 3D printing allows for the creation of porous structures within the titanium bone screws. This "Trabecular Titanium" mimics the natural bone architecture, significantly improving long-term stability.
With the rise of hand and foot surgeries, the industry is moving towards smaller, high-strength screws. Our R&D team is focused on producing screws with diameters as small as 1.5mm while maintaining structural integrity under high stress.
Understanding the local application of Titanium Bone Screws is vital for any global exporter. In developed markets like North America and Europe, the focus is heavily on trauma centers and specialized orthopedic hospitals where high-value Herbert Screws and Cannulated Screws are frequently used for scaphoid fractures and intra-articular injuries.
In contrast, emerging markets in Southeast Asia and Africa often prioritize cost-effective yet high-quality universal bone screws for general trauma care. As a versatile manufacturer, we tailor our production lines to meet these varied needs, ensuring that our implants perform flawlessly whether in a high-tech robotic surgery suite in London or a rural trauma clinic in Brazil.
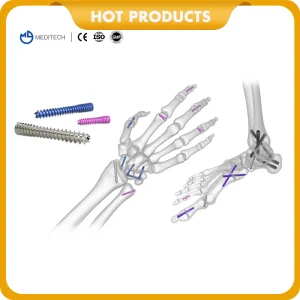
From raw material sourcing of medical-grade titanium to final sterile packaging, our factory maintains full control. This ensures a consistent supply chain and the ability to scale production rapidly for large-scale tender orders.
Our quality control philosophy is "Quality and integrity first, striving for excellence." Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed international quality and safety standards, including CE and FDA certifications.
By leveraging the industrial ecosystem in China, we offer high-end medical devices at a price point that provides significant value to distributors and hospitals worldwide without compromising on patient safety.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.

The mechanical properties of titanium alloys are unparalleled in orthopedic surgery. As a Titanium Bone Screws exporter, we often explain to our clients that Titanium Ti-6Al-4V offers a unique combination of high fatigue strength and a low elastic modulus. The latter is particularly important because it is closer to that of human bone compared to stainless steel, which helps in reducing "stress shielding"—a phenomenon where the implant takes too much load, causing the surrounding bone to weaken over time.
Furthermore, the chemical stability of titanium means that it does not release toxic ions into the body, preventing allergic reactions and inflammatory responses. This biocompatibility is the reason why titanium has become the preferred material for long-term implants. Our manufacturing process involves precise CNC grinding and thread rolling to ensure that every screw has a smooth finish, reducing tissue irritation and facilitating easier removal if necessary after the bone has healed.
Looking at the global procurement trends, we see a massive rise in demand for "Universal Trauma Sets." These sets include a variety of titanium screws—locking, non-locking, cortical, and cancellous—all organized in a single tray for surgical efficiency. As a factory, we have adapted by offering customizable kit configurations, allowing hospitals to stock exactly what they need for their specific patient demographics.
In the industrial context, the "Top 10" status of a supplier is defined by their ability to maintain tight tolerances. Our bone screws are manufactured to micron-level precision. This level of accuracy is essential for the interface between the screw head and the screwdriver, ensuring that "stripping" does not occur during the high-torque environment of an operation. We utilize advanced optical inspection systems to verify every batch before it leaves our facility.
Finally, the environmental and economic impact of sourcing from a Chinese factory like ours cannot be overstated. With the most robust manufacturing ecosystem in the world, we can pivot to new materials and designs faster than Western counterparts. Whether it is moving from Grade 2 to Grade 5 titanium or implementing a new double-lead thread design, our response time to market needs is what keeps us at the forefront of the global orthopedic industry.