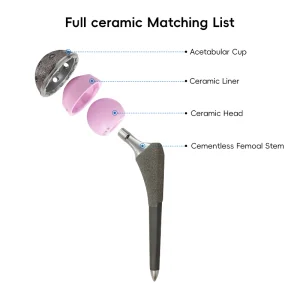

Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement

Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail


Titanium Orthopedic Femur Interlocking Nail Medical Bone Surgical Femoral Intramedullary Implant


Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
The global orthopedic implant market is witnessing an unprecedented transformation, with femoral head technology at its core. As a critical component of total hip arthroplasty (THA), the demand for high-performance femoral heads has surged globally. This growth is driven by a demographic shift towards an aging population and an increase in orthopedic conditions such as osteoarthritis and femoral neck fractures.
In the current industrial landscape, the competition among OEM/ODM Femoral Head manufacturers has shifted from simple mechanical production to high-precision material science. Manufacturers are now required to integrate advanced robotics, AI-driven quality inspection, and biocompatible material innovation to meet the rigorous demands of modern surgery.
Our products are exported to over 70 countries, bridging the gap between high-end medical technology and affordable healthcare solutions.
Specializing in Medical Titanium and Cobalt-Chrome alloys, ensuring maximum longevity and reduced wear for patients.
Every femoral head undergoes rigorous AI-assisted visual inspections to ensure zero-defect manufacturing.
As a leading Femoral Head exporter, we stay ahead of the curve by monitoring key trends that define the future of orthopedics:
Global medical device distributors and hospital chains are increasingly seeking partners who can provide more than just a product. The modern procurement landscape demands:
1. Strict Regulatory Compliance: Requirements for ISO 13485, CE, and FDA certifications are non-negotiable for entering regulated markets in Europe and North America.
2. Supply Chain Resilience: The ability for a factory to maintain consistent output despite global logistics challenges.
3. Customization (OEM/ODM): The flexibility to modify femoral stem lengths, head diameters, and taper designs to fit local anatomical variations (e.g., Asian vs. Caucasian bone structures).
Located at the heart of China's medical industrial cluster, our factory combines cost-efficiency with uncompromising quality. We bridge the gap between "Made in China" and "World-Class Quality."
Export Countries
13485 Certified
Years Experience
Quality Inspected
As a dedicated OEM/ODM Femoral Head manufacturer, we offer a comprehensive suite of services from initial CAD design to sterile packaging. Our R&D team works directly with orthopedic surgeons to refine designs that solve real-world clinical challenges.
Our Quality Control Philosophy: "Quality and integrity first, striving for excellence, and pursuing the highest standards." This is not just a slogan; it is the blueprint of our production line.

Our Commitment to Excellence and Innovation
Understanding the local context is vital. We provide specialized solutions for different medical environments:
Our professional R&D team maintains long-term technical exchanges with leading hospital experts. This ensure our products function to their fullest potential in the operating room. Whether it's a Titanium Cemented Femoral Stem or a Proximal Femoral Intramedullary Nail, each product is meticulously crafted to ensure anatomical compatibility and ease of implantation.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product. Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications.
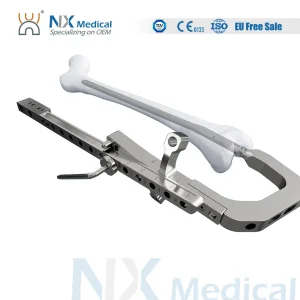

Nx Medical Orthopedic Retrograde Femoral Intramedullary Interlocking Nail System for Trauma Implant

Artificial Knee Implant Cementless Femoral Stem Knee Replacement Instrument Kit


Advanced Trochanteric Femoral Intramedullary Nail for Fracture Repair



Medical Titanium Bone Surgical System Pfna Orthopedic Femoral Interlocking Intramedullary Nail
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.
Contact us today to discuss your OEM/ODM requirements and join our network of successful global partners.