The global market for Cranial Mesh and neurosurgical implants is witnessing an unprecedented era of growth, driven by the increasing prevalence of neurological disorders, traumatic brain injuries (TBIs), and the rising demand for reconstructive surgeries. As of 2024, the industry has transitioned from standardized off-the-shelf plates to highly sophisticated, biocompatible Grade 5 Titanium and PEEK (Polyether ether ketone) solutions.
Modern neurosurgery demands precision. The global healthcare sector is shifting towards Patient-Specific Implants (PSI), where digital twins of a patient's skull are created using high-resolution CT scans. Chinese manufacturers have emerged as pivotal players in this landscape, bridging the gap between high-end technological innovation and cost-effective production, making life-saving surgeries accessible across both developed and emerging markets.
Rapid adoption of 3D printing (Additive Manufacturing) allows for intricate porous structures that facilitate better osseointegration and reduce post-operative complications.
While Titanium remains the gold standard for its strength-to-weight ratio, PEEK implants are gaining traction for their radiolucency, allowing clearer MRI/CT imaging post-surgery.
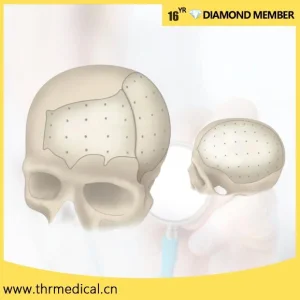
The focus has shifted to "anatomical fitting," where the mesh contour perfectly matches the skull defect, reducing operation time by up to 30%.
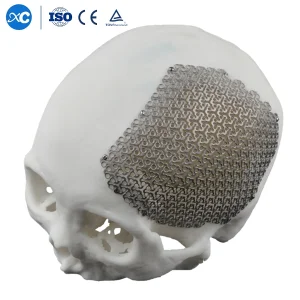
Our flagship 3D Anatomical Cloud Shape CMF Cranial Mesh represents the pinnacle of reconstructive technology. Engineered for complex maxillofacial and neurosurgical repairs, this implant features a unique "cloud" geometry that allows for superior flexibility during fixations while maintaining rigid protection for the cerebral cortex.
Procurement departments globally prioritize this model due to its versatility in treating varied fracture patterns and its pre-contoured design which minimizes the need for intraoperative bending.
International hospital groups and medical device distributors look for three core criteria: Compliance, Customization, and Cost-efficiency. In regions like North America and Europe, the demand is heavily focused on regulatory clearance (FDA/CE) and the ability to integrate with surgical navigation systems. In Southeast Asia and Africa, the focus remains on high-quality, durable trauma fixation sets that can be delivered quickly for emergency interventions.
For surgeons requiring the ultimate in personalization, the Surgical PEEK Patient Specific Implant offers a metal-free alternative. This system is custom-milled to the exact dimensions of the patient's defect, ensuring a seamless "lock and key" fit.
The PEEK material's thermal conductivity is similar to natural bone, reducing patient sensitivity to temperature changes—a common complaint with traditional metal meshes. It is the preferred choice for high-end private clinics and specialized neurology centers.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential.
Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.

From raw titanium sponge processing to sterile packaging, our integrated supply chain ensures full traceability and competitive lead times.
We iterate designs based on feedback from global neurosurgeons, allowing us to release updated cranial mesh patterns faster than Western competitors.
By optimizing manufacturing efficiency, we provide medical-grade implants at a fraction of the cost, assisting healthcare systems in managing budget constraints.