




1 / 5










| Still deciding? Get samples of $ ! US$ 22/Piece Request Sample |
Legerity 5.5 Posterior Thoracolumbar Fixation System
Model: DFⅡ
Indications:
Implants Features:



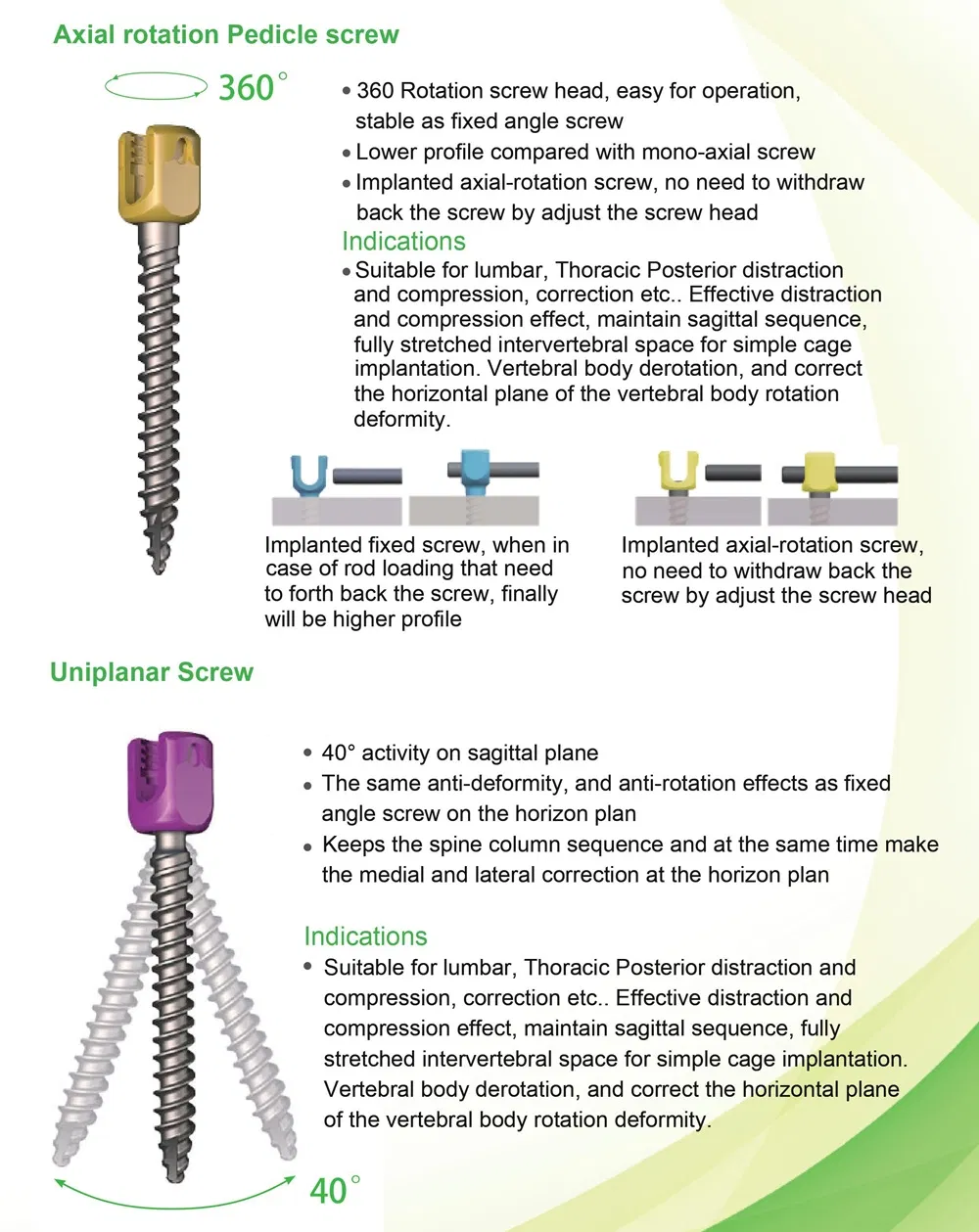
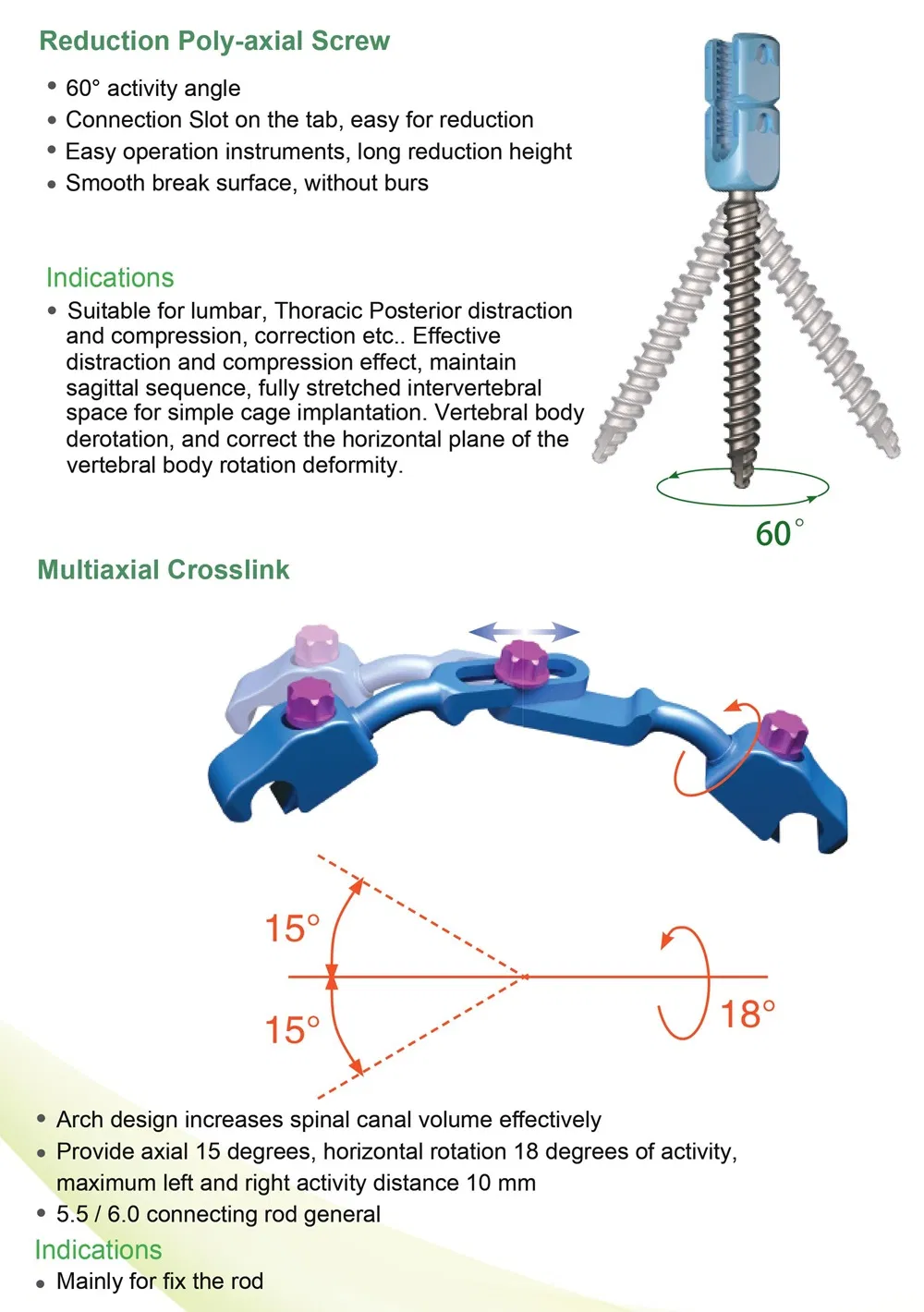
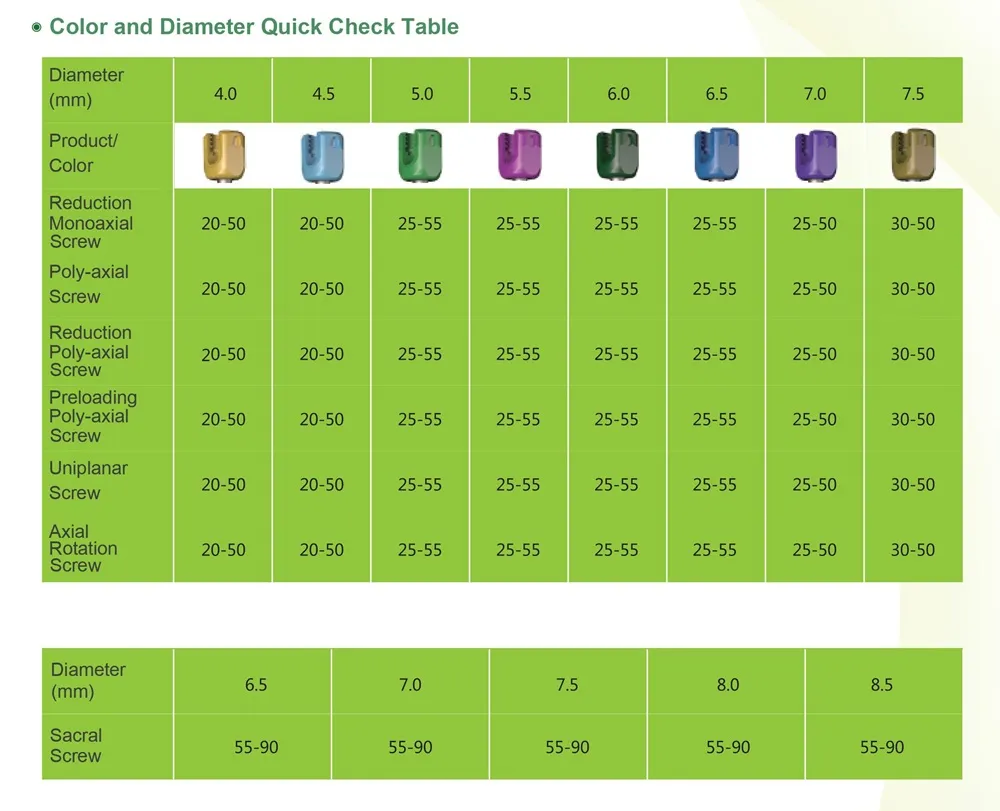


Packing Details:
Delivery Way:

Capacity & Expertise:

Factory Advantages:


 Daher Orthopedic Implants
Daher Orthopedic Implants