


1 / 5










| Still deciding? Get samples of $ ! US$ 1600/Set Request Sample |
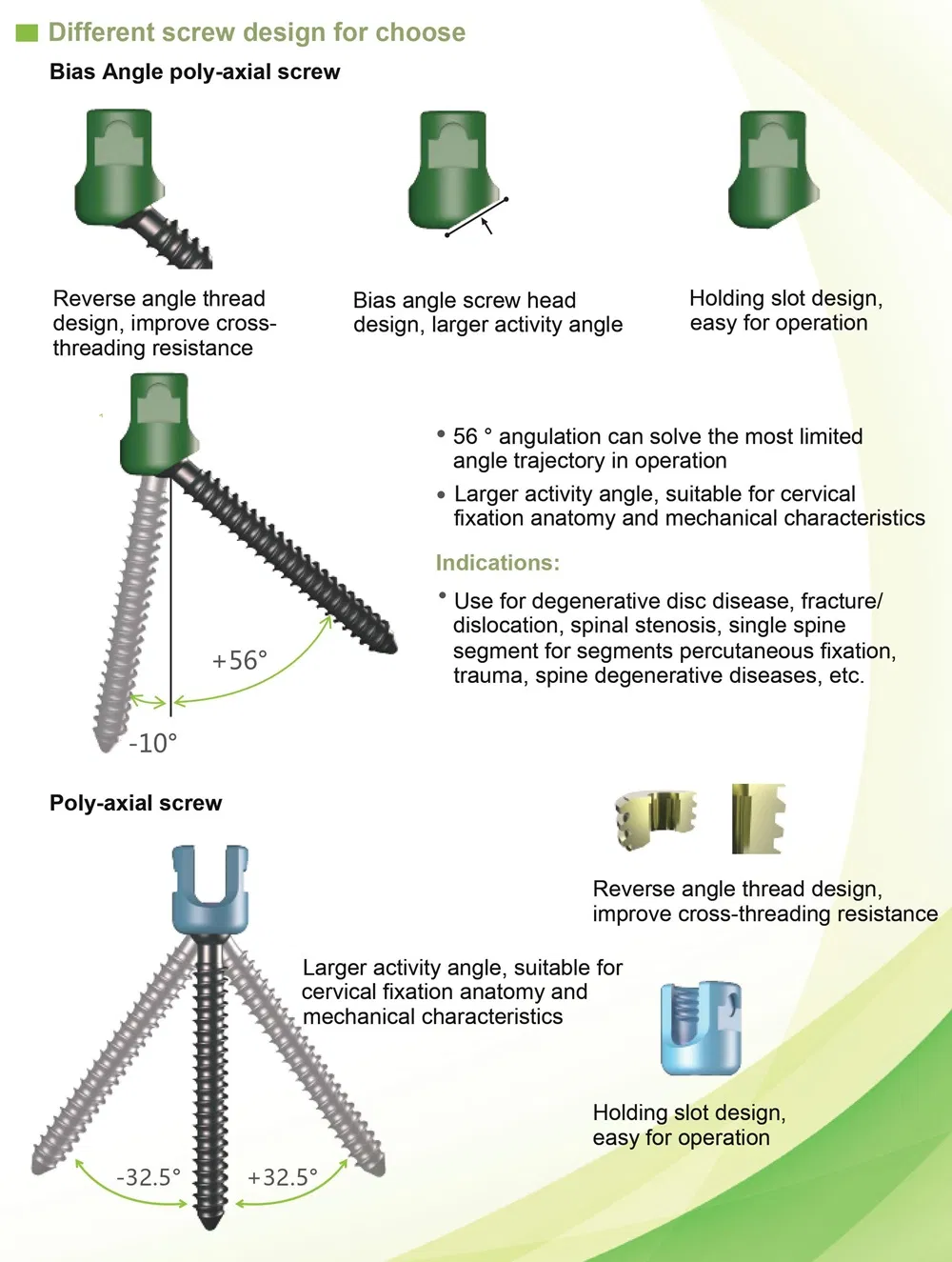
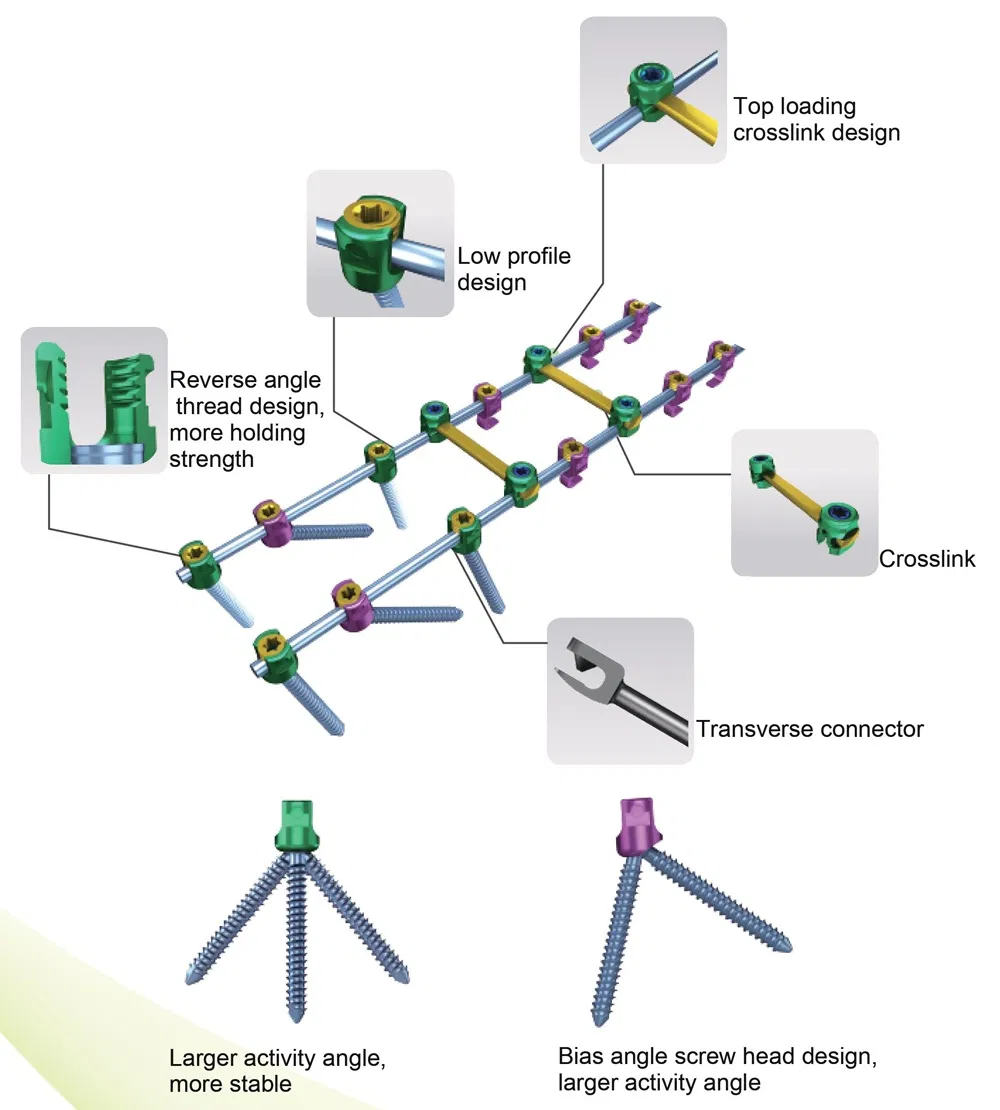
Key Features:
Clinical Applications:
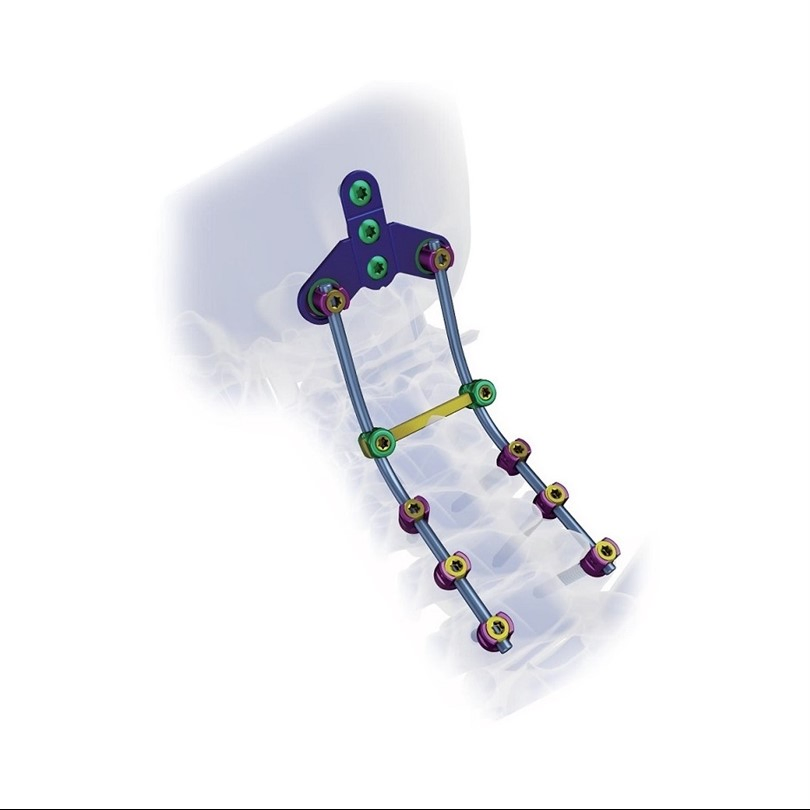
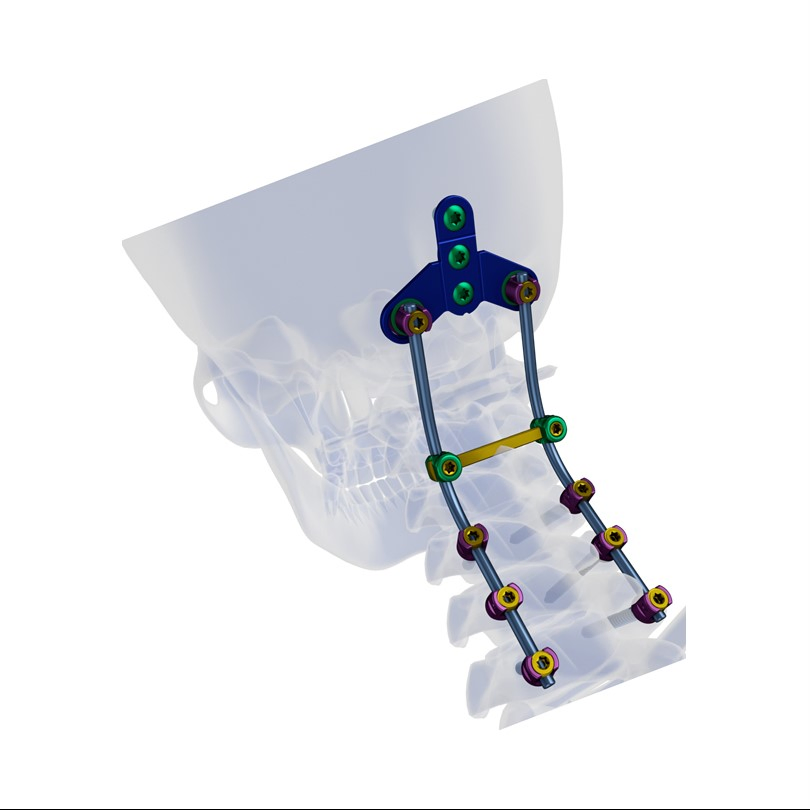
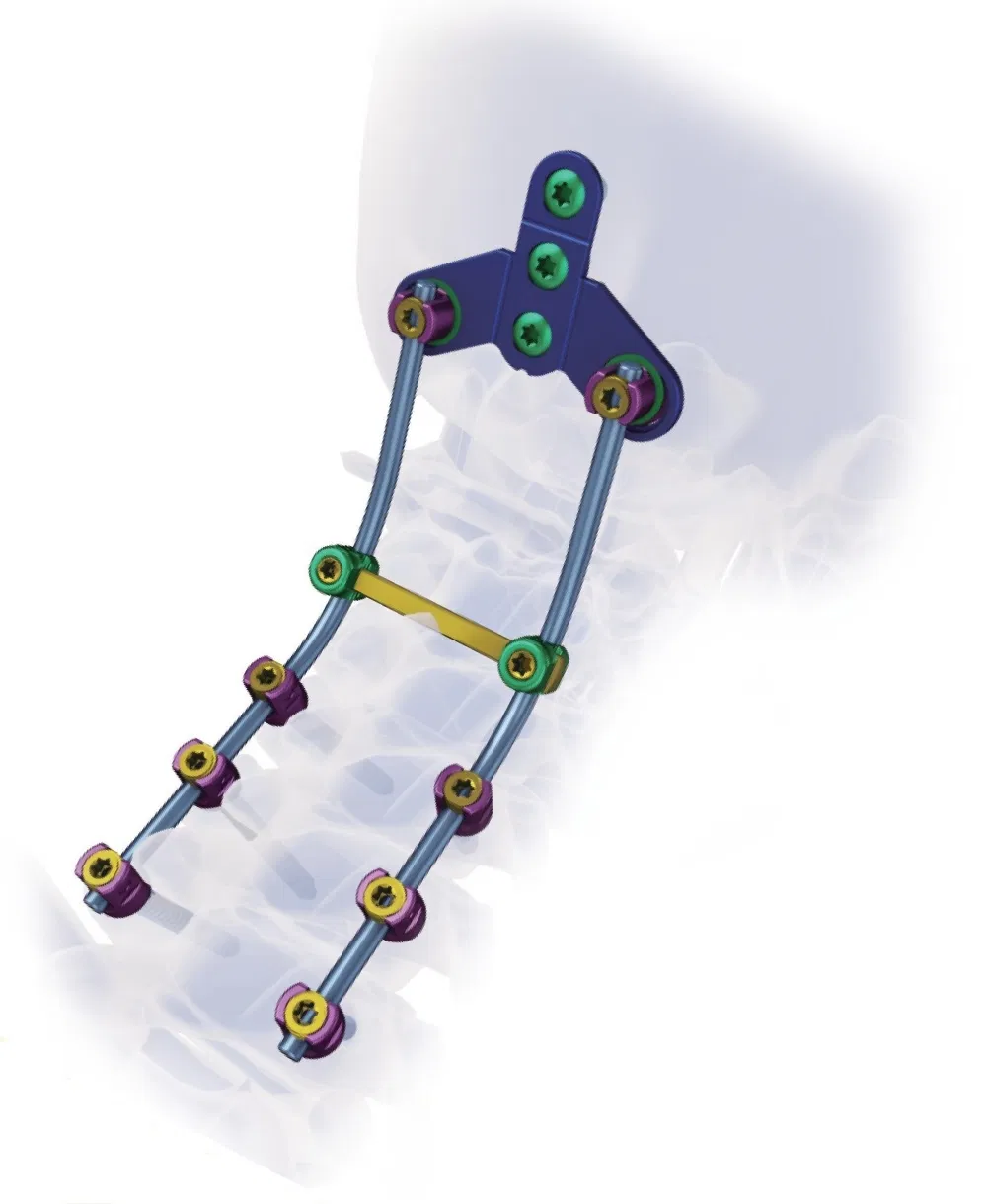
Superior biocompatibility, ideal for treating degenerative disc diseases, fractures, dislocations, spinal stenosis, and occipital-cervical disassociation.


Packing Details:
Delivery Options:

Focus & Capacity: One-stop orthopedic product solution provider since 2016, with a monthly capacity of 10,000 units.
Infrastructure:



 Daher Orthopedic Implants
Daher Orthopedic Implants