




1 / 5










| Still deciding? Get samples of $ ! US$ 1.2/Piece Request Sample |
Designed for internal fixation of fractures, osteotomies, and non-unions, particularly in small bone and joint surgery. The cannulated design allows insertion over a guide wire for precise placement.

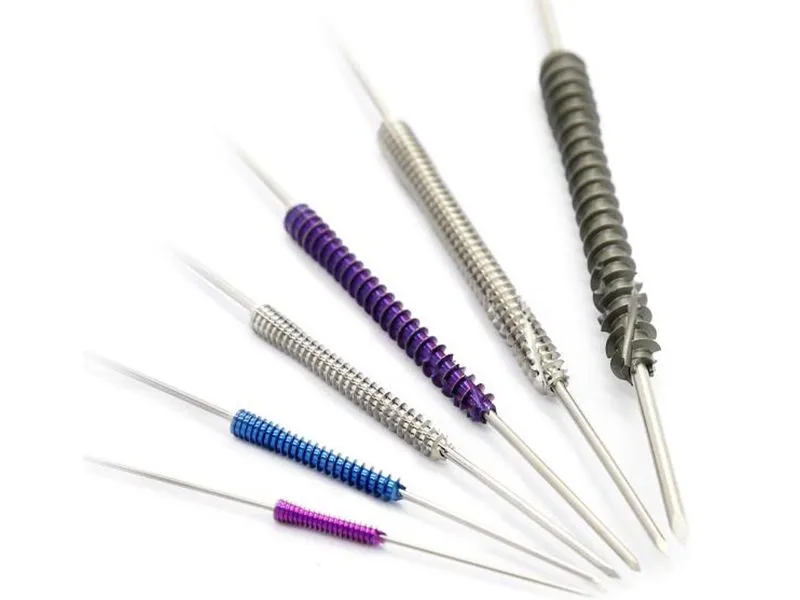

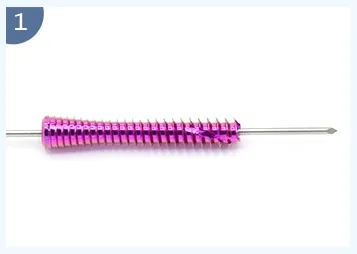



| Product | REF (Titanium) | Specification |
|---|---|---|
| Cannulated Headless Compression Screw | T4100-(9501-9510) | 3.0*(12-30mm) |
| T4100-(9511-9524) | 3.5*(14-40mm) | |
| T4100-(9525-9544) | 4.0*(16-60mm) | |
| T4100-(9545-9560) | 5.0*(20-50mm) | |
| T4100-9562 | 5.0*60mm | |
| T4100-9564 | 5.0*70mm |



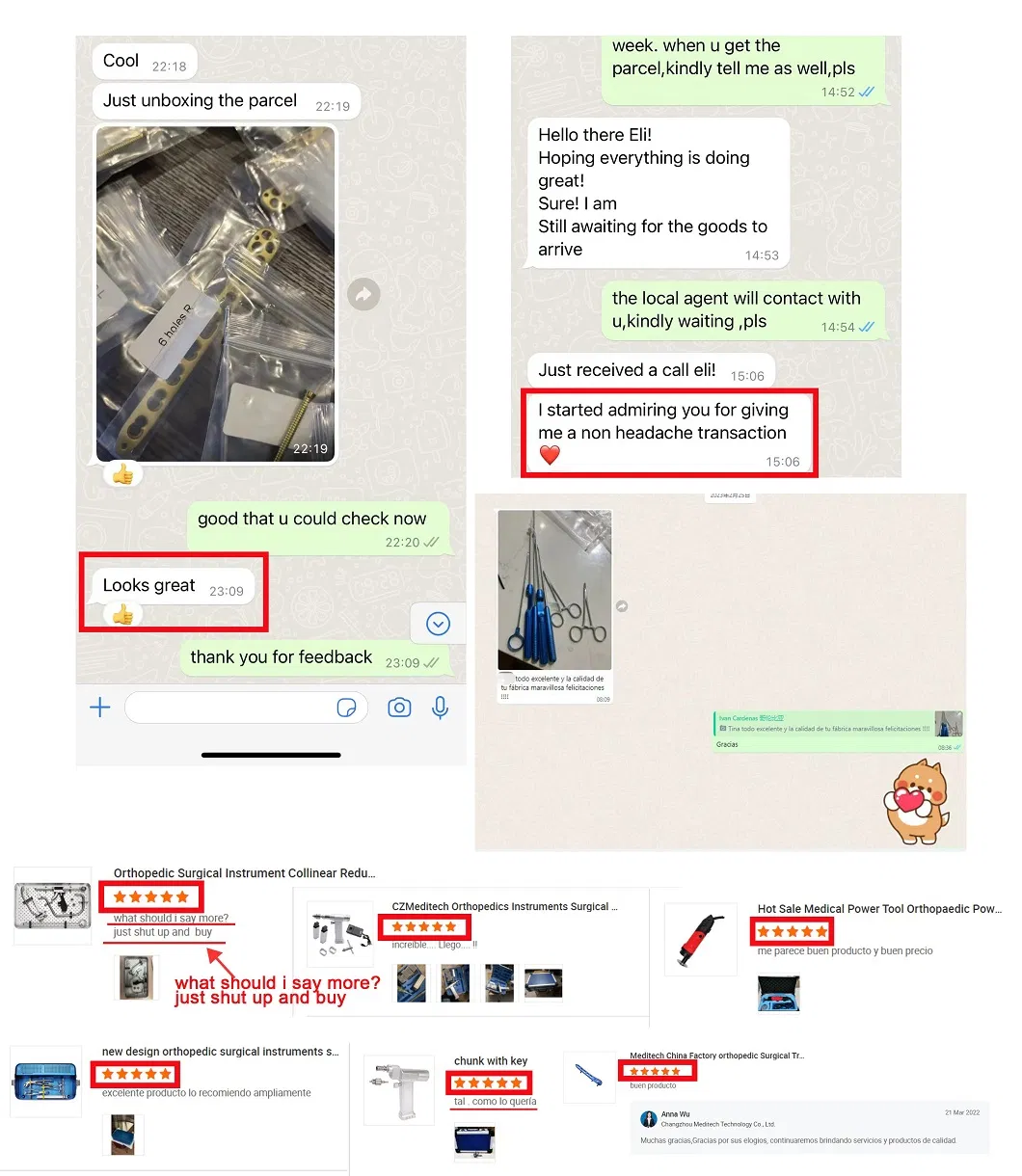
As a leader in orthopedic implants and instruments manufacturing, we have successfully supplied to 2,500+ clients in 70+ countries for over 14 years. With cutting edge equipment, we offer products of the highest industrial standards. We are continually pushing the limits of our know-how to provide high-quality, innovative product solutions for human health worldwide.

 Daher Orthopedic Implants
Daher Orthopedic Implants