🏢 Company Profile
Established in 1958, we are engaged in the development, manufacture and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus, the product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis and surgical instruments.
Our products are characterized with multi-varieties and complete specifications. All apparatus have passed the approval of national and provincial superintendent offices. Advanced equipment including imported CNC and high precision digital control machines, as well as a 100,000 Grade axenic purification chamber, contribute to high product quality.
We are the first to pass ISO 9001 quality system certification in our sector. Our brand has won national silver awards and invention prizes. We adhere to the tenet of "create high quality with science and technology, serve the society with love."
❓ Frequently Asked Questions
What types of orthopedic products do you manufacture?
We specialize in a wide range of orthopedic solutions including metal bone plates, screw systems, interlocking intramedullary nails, spinal fixation devices, and external fixation systems.
How is the quality of the medical devices guaranteed?
All products are produced according to EU CE standards. We maintain a strict quality control system with ISO and GMP certificates, including pre-production samples and final inspections before shipment.
What is the experience level of your manufacturing facility?
Our facility was established in 1958, bringing over 60 years of expertise in R&D, manufacturing, and international sales of orthopedic implants and instruments.
Can you handle custom OEM or ODM requests?
Yes, we accept OEM and ODM orders to meet specific surgical requirements and regional market needs.
What are the standard shipping methods and terms?
We offer various delivery terms including FOB, CIF, and EXW via major carriers like DHL, UPS, FedEx, and TNT Air Cargo. Payment is accepted via T/T, L/C, and Western Union.
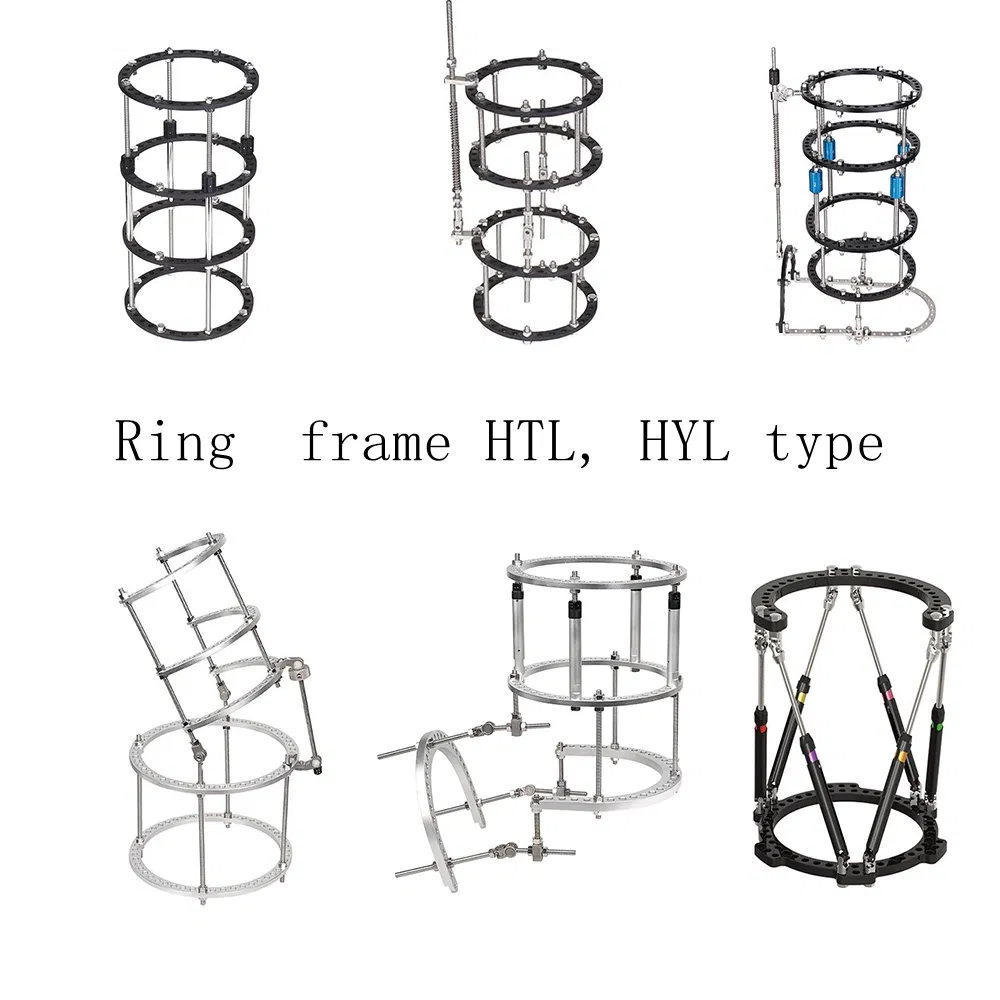
Who is the primary target group for these fixators?
These specific external fixators (Model HYL004) are designed primarily for adult use in complex bone fracture fixation and deformity correction.





















 Daher Orthopedic Implants
Daher Orthopedic Implants