 Surgical Instrument Set Medical Equipment Orthopedic Acl Instrument
Surgical Instrument Set Medical Equipment Orthopedic Acl Instrument
 Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
 Instruments for Knee Replacement Surgery Artificial Hip Joint System Hip Joint Replacement Prosthesis
Instruments for Knee Replacement Surgery Artificial Hip Joint System Hip Joint Replacement Prosthesis
 Replacement Sterilized Package Prosthesis Knee Joint World′ S Leading 3D Printed
Replacement Sterilized Package Prosthesis Knee Joint World′ S Leading 3D Printed
Denmark has long been recognized as a global leader in life sciences and medtech innovation. As a premier Revision Hip/Knee Systems Supplier & Exporter in Denmark, we understand that the Danish healthcare system (Sundhedsvæsenet) demands the highest level of clinical evidence and engineering precision. With a robust infrastructure centered around world-class institutions like Rigshospitalet and Aarhus University Hospital, Denmark is at the forefront of adopting advanced arthroplasty solutions.
The industrial status of orthopedic implants in Denmark is characterized by a shift toward value-based healthcare. Hospitals across the Capital Region (Region Hovedstaden) and Central Denmark Region are increasingly seeking revision systems that reduce operating room time and improve long-term patient outcomes. Revision surgeries—procedures performed to replace or compensate for a failed primary implant—are technically demanding. Our role as a supplier is to provide the Danish medical community with modular, adaptable, and highly biocompatible systems that address complex bone loss and mechanical instability.
Our systems are designed to mimic natural human kinematics, ensuring that revision patients in Denmark regain mobility faster with reduced risk of dislocation.
Utilizing computational modeling to simulate stress distribution in titanium alloy implants, ensuring durability for decades of use.
All products meet the rigorous safety requirements of the Danish Medicines Agency and the latest EU Medical Device Regulations (MDR).
The orthopedic industry in Denmark is witnessing a transformative era driven by digital health and material science. One of the most significant trends is the integration of 3D-printed porous structures. Unlike traditional implants, these 3D-printed components allow for superior osseointegration, which is critical in revision cases where bone quality may be compromised.
Furthermore, the "Green Transition" in Denmark’s industrial sector is influencing medical manufacturing. As a responsible exporter, we are optimizing our production processes to align with Denmark’s sustainability goals, focusing on sterilized, eco-friendly packaging and long-lifecycle instrumentation. The rise of Robotic-Assisted Surgery (RAS) in Danish clinics also requires revision systems to be compatible with digital navigation tools, a requirement our latest instrument sets meet with sub-millimeter precision.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product. Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential. Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.

In Danish clinical practice, revision hip and knee surgeries often arise from aseptic loosening, periprosthetic infection, or fractures. Our S-ROM Titanium Alloy Cementless systems are particularly popular in Nordic regions due to their modularity, allowing Danish surgeons to customize the implant fit to the patient’s unique anatomy intraoperatively.
Scenario A: The Copenhagen Case. In high-volume orthopedic centers in Copenhagen, the use of our Disposable Pulsed Lavage System is critical for thorough debridement during revision surgery, significantly reducing the risk of post-operative infection—a key performance indicator for Danish health boards.
Scenario B: Geriatric Care in Odense. With Denmark’s aging population, revision knee systems must account for severe bone loss. Our 3D-printed replacement joints provide a biological fix that traditional cemented implants cannot match, promoting faster recovery for elderly Danish citizens and reducing the burden on the social care system.
 Prosthesis Replacement Orthopedic Sterilized Package Class III Knee Joint
Prosthesis Replacement Orthopedic Sterilized Package Class III Knee Joint
 S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
 Disposable Pulsed Lavage System
Disposable Pulsed Lavage System
 Medical Orthopedic Spine Endoscopy Instrument Set Surgery Used
Medical Orthopedic Spine Endoscopy Instrument Set Surgery Used
 Hospital Equipment Retractor Instrument Set Orthopedic Surgery
Hospital Equipment Retractor Instrument Set Orthopedic Surgery
 Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
 Hip Joint Prosthesis Replacement Knee Joint Acetabular Cup
Hip Joint Prosthesis Replacement Knee Joint Acetabular Cup
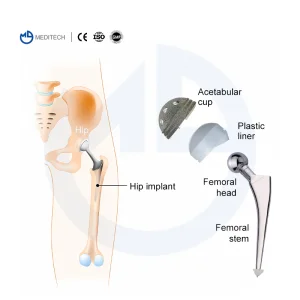 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
As a global exporter to Denmark, we provide more than just implants; we provide comprehensive surgical solutions. Our instrument sets, including the Orthopedic Surgical Instrument Set for Orthopaedic Cable Instrument and Medical Endoscope Instruments Set, are designed for ergonomics and longevity. In the fast-paced environment of a Danish Operating Theatre (OP), instruments must be intuitive and easy to sterilize.
Our revision hip systems utilize advanced Acetabular Cup designs that offer multiple screw hole options for secure fixation in deficient bone. For knee revisions, our TKA (Total Knee Arthroplasty) systems provide various constraint levels, from posterior-stabilized to fully constrained hinges, allowing surgeons in Denmark to address the full spectrum of ligamentous instability.
The manufacturing process involves state-of-the-art CNC machining and proprietary surface treatments that enhance the bio-affinity of the titanium alloy. By maintaining a strict quality control loop, we ensure that every product arriving in Denmark—whether it's a sterile-packaged knee joint or a complex endoscope set—performs flawlessly in the most demanding surgical environments.
Procuring medical devices in the Danish market requires a partner who understands the Amgros procurement system and the technical requirements of local regional tenders. We offer full documentation support, including clinical evaluation reports (CER) and stability testing data, ensuring a smooth registration and supply process for our Danish partners.
Connect with our technical team today for a customized quote and clinical consultation.
Send Inquiry Now