 Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
 High Quality Orthopedic Titanium Metal Cancellous Cannulated Screws Stainless Steel Full-Thread Self-Tapping Bone Screw
High Quality Orthopedic Titanium Metal Cancellous Cannulated Screws Stainless Steel Full-Thread Self-Tapping Bone Screw
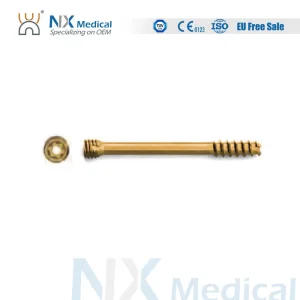 Nx Medical Orthopedic Implant Cannulated Headless Compression Screw Metallic Bone Screws
Nx Medical Orthopedic Implant Cannulated Headless Compression Screw Metallic Bone Screws
 Titanium Scew, Headless Screw, Compression Screws, Surgical Instrument, Cannulated Screw
Titanium Scew, Headless Screw, Compression Screws, Surgical Instrument, Cannulated Screw
01 Chile has emerged as one of the most sophisticated healthcare markets in Latin America. With a dual system comprising the public FONASA and private ISAPRE networks, the demand for high-quality orthopedic implants, specifically Cannulated Screws, has seen a steady 15% annual growth. From Santiago to Concepción, clinics are prioritizing minimally invasive procedures that require the precision of hollow-core screw technology.
As a leading supplier, we understand that Chilean medical institutions like Clinica Alemana and Hospital Clínico de la Universidad de Chile demand instruments that meet rigorous international standards. Our cannulated screws are designed to facilitate guide-wire insertion, ensuring that complex fractures are stabilized with minimal surgical trauma.
The industrial climate in Chile is currently focused on Economic Efficiency and Patient Outcomes. The Chilean Institute of Public Health (ISP - Instituto de Salud Pública) has set high benchmarks for medical imports. Our manufacturing process aligns perfectly with these requirements, offering:
Our screws utilize Ti-6Al-4V ELI, providing the perfect balance of strength and flexibility required for the diverse patient demographics in Chile, from high-altitude mining workers in the North to the active urban population in Santiago.
Reducing surgery time is critical in Chile's busy public hospitals. Our self-tapping designs eliminate extra steps, allowing for faster fixation and reduced anesthesia time.
In the Chilean context, our cannulated screws are widely applied in several specific medical fields:

We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential.
Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.
| Screw Type | Diameter Range | Main Application | Chilean Standard Compliance |
|---|---|---|---|
| Micro Cannulated | 2.0mm - 3.0mm | Hand & Foot Surgery (Santiago Trauma Clinics) | CE / ISO 13485 / ISP Chile |
| Small Cannulated | 3.5mm - 4.5mm | Ankle & Patella Fractures | CE / ISO 13485 / FDA |
| Large Cannulated | 6.5mm - 7.3mm | Femoral Neck (Hip Fractures) | ASTM F136 Compliant |
As a global supplier, we offer optimized logistics to Chile. Whether your facility is located in Valparaíso, Antofagasta, or the Magallanes region, we ensure that our cannulated screws reach you through reliable air freight and sea routes, handling all documentation required for Chilean customs clearance.
 Hb7.3 Titanium or Ss Cannulated Compression Screws
Hb7.3 Titanium or Ss Cannulated Compression Screws
 Interference Screws for Acl Reconstruction Peek and Cannulated Screws Sports Medicine
Interference Screws for Acl Reconstruction Peek and Cannulated Screws Sports Medicine
 Manufacturer Wholesale Professional High Quality 3.0 Orthopaedic Compression Cannulated Screw
Manufacturer Wholesale Professional High Quality 3.0 Orthopaedic Compression Cannulated Screw
 Nx Medical Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5 for Bone Fixation
Nx Medical Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5 for Bone Fixation
 Factory Direct Supply Orthopedic Spine Implant Bone Cement-Injectable Cannulated Pedicle Screws High Quality
Factory Direct Supply Orthopedic Spine Implant Bone Cement-Injectable Cannulated Pedicle Screws High Quality
 Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
 Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
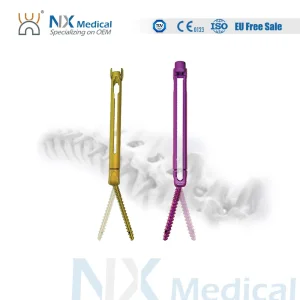 Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw Spinal Fixation
Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw Spinal Fixation
The future of Cannulated Screw technology in Chile lies in the integration of 3D-planned surgical guides and patient-specific implant sets. As a leading factory and supplier, we are already developing next-generation bone cement injectable screws to address the specific needs of the osteoporotic patient demographic in Chile.
Whether you are a distributor in Santiago, a purchasing officer for a Metropolitan Health Service, or a private clinic owner, our commitment is to provide the highest quality implants that ensure the rapid recovery of Chilean patients.