Anterior Lumbar Interbody Fusion (ALIF) has revolutionized the way surgeons approach degenerative disc diseases and spinal instability. As a leading China Top ALIF Cages manufacturer, we recognize that the global market for interbody fusion devices is currently experiencing an unprecedented surge. This growth is driven by an aging population, an increase in sports-related spinal injuries, and the rising demand for minimally invasive surgical (MIS) procedures that offer faster recovery times and less postoperative pain.
In today's commercial environment, medical institutions in North America, Europe, and the Asia-Pacific region are prioritizing implants that offer superior primary stability and long-term osteointegration. ALIF Cages, specifically those engineered with advanced materials like PEEK (Polyetheretherketone) and Titanium-coated surfaces, have become the gold standard. They provide a larger surface area for fusion compared to posterior approaches, allowing for better restoration of lumbar lordosis and disc height.
International procurement trends show a shift towards multi-functional cages that support both traditional and robotic-assisted surgeries. Hospitals are seeking suppliers who can provide consistent quality across large volumes.
The integration of AI-guided design and 3D printing in the manufacturing process ensures that each ALIF cage meets precise anatomical requirements, reducing the risk of cage subsidence.
As global standards tighten, being a manufacturer with ISO 13485, CE, and FDA compliance is no longer an option—it is a prerequisite for entry into the high-end medical market.
The orthopedic industry is moving towards "Bio-Intelligence." We are at the forefront of this movement by developing Titanium Coating Bullet Lumbar PEEK Interbody Fusion Cages. Why is this significant? While PEEK offers a modulus of elasticity similar to human bone (reducing stress shielding), its hydrophobic nature can limit bone attachment. By applying a plasma-sprayed titanium coating, we combine the mechanical advantages of PEEK with the excellent osteoconductive properties of titanium.
Furthermore, the trend towards 3D-printed porous structures is gaining momentum. These structures mimic the architecture of trabecular bone, promoting rapid bone ingrowth throughout the implant. As a specialist supplier, our R&D team is constantly exploring these frontiers to ensure our clients receive the most advanced medical solutions available.
Choosing a China Top ALIF Cages supplier offers unique advantages in the global supply chain. Our facility in China leverages a highly sophisticated manufacturing ecosystem, allowing us to maintain rigorous quality control while optimizing production costs. This cost-efficiency is passed directly to our global partners, enabling hospitals and distributors to provide high-quality care without the prohibitive price tags often associated with Western brands.
Our "Smart Factory" approach utilizes automated CNC machining and clean-room packaging to ensure that every orthopedic implant—from ALIF cages to locking compression plates—is sterile, precise, and ready for the operating room. We don't just manufacture; we innovate through "Long-term technical exchanges with leading hospital experts," ensuring our designs solve real-world surgical challenges.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential.
ALIF cages are utilized in various clinical scenarios globally:
For global procurement managers, the decision-making process involves balancing regulatory safety with supply chain reliability. Our factory addresses these needs by providing comprehensive documentation, including ISO 9001 and ISO 13485 certifications. Our products exceed international quality and safety standards, including CE and FDA certifications, making them suitable for the most demanding medical markets in Europe and America.
Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.


Titanium Medical Orthopedic Trauma Radius Bone Fracture Reconstruction LCP Locking Compression Plate

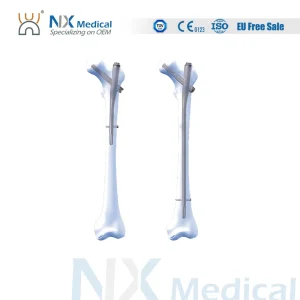
Nx Medical Orthopedic Implant Proximal Femoral Interlocking Intramedullary Nail
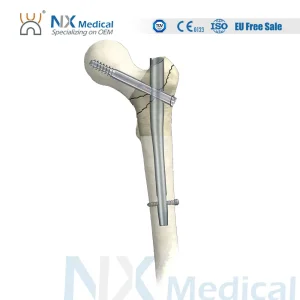
Nx Medical Orthopedic Trauma Titanium Intramedullary Interlocking Femoral Femur Pfna Bone Nail
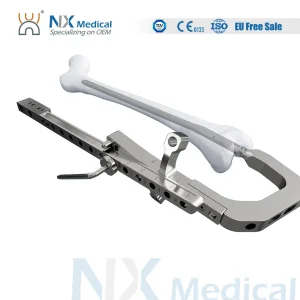

China Factory Nx Medical Orthopedic Trauma Implant Titanium Retrograde Tibial Intramedullary Nail

Nx Medical Orthopedic Retrograde Femoral Intramedullary Interlocking Nail System for Trauma Implant
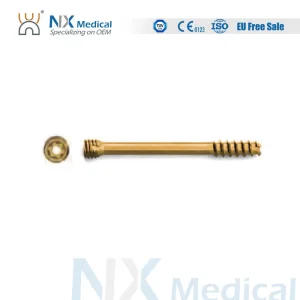
Nx Medical Orthopedic Implant Cannulated Headless Compression Screw Metallic Bone Screws
The selection of an interbody fusion cage is a critical decision in spinal surgery. As a China Top ALIF Cages manufacturer, we understand that the implant is not just a piece of hardware; it is a life-changing device for the patient. The anterior lumbar interbody fusion (ALIF) approach allows surgeons to place a larger cage, which provides a significant increase in stability and fusion surface area. This is particularly crucial in multi-level fusion procedures where the risk of pseudoarthrosis is higher.
Our commitment to excellence is reflected in our material choices. PEEK (Polyetheretherketone) remains a favorite among surgeons due to its radiolucent properties, allowing for easy monitoring of bone growth via X-ray. However, our Titanium Coating technology elevates this by providing an interface that bone cells "recognize" and attach to more readily. This "best of both worlds" approach is a cornerstone of our research and development, ensuring that our ALIF Cages meet the biological and mechanical needs of the human body.
We invite global distributors, hospital procurement departments, and orthopedic surgeons to partner with us. Our factory is equipped to handle OEM/ODM requests, allowing for localized adaptations to meet specific regional clinical requirements. Whether you are looking for a reliable supply of bullet lumbar fusion cages or advanced trauma locking plates, our professional team is ready to provide the technical expertise and high-quality products your patients deserve.
In conclusion, the intersection of AI-driven design, advanced material science, and cost-effective Chinese manufacturing makes us the ideal partner in the competitive field of orthopedic implants. We continue to strive for excellence, ensuring that every screw, plate, and cage that leaves our facility is a testament to our quality control philosophy: "Quality and integrity first."