











| Customization: | Available |
|---|---|
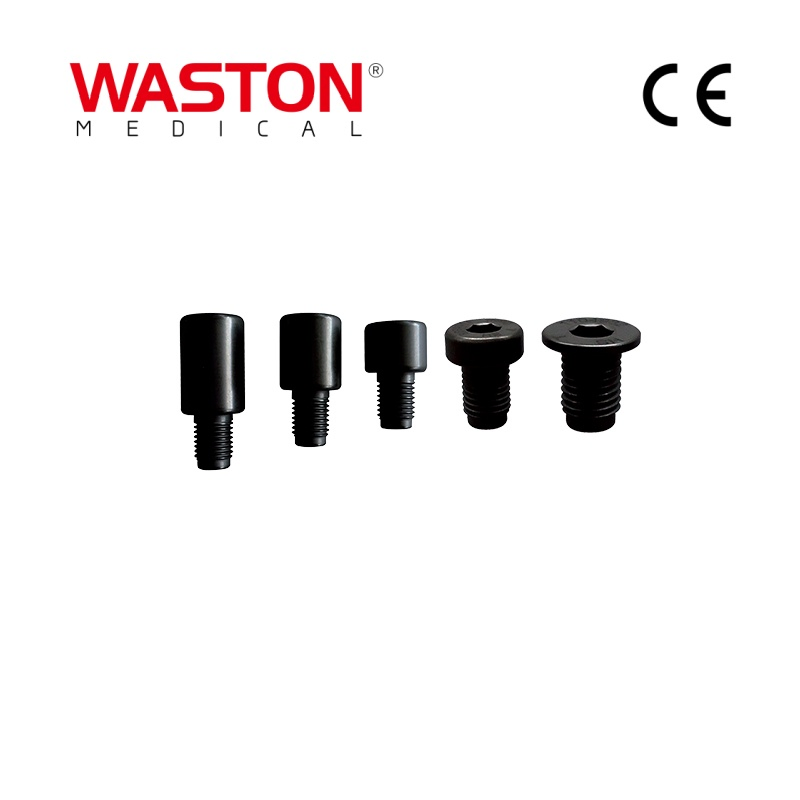
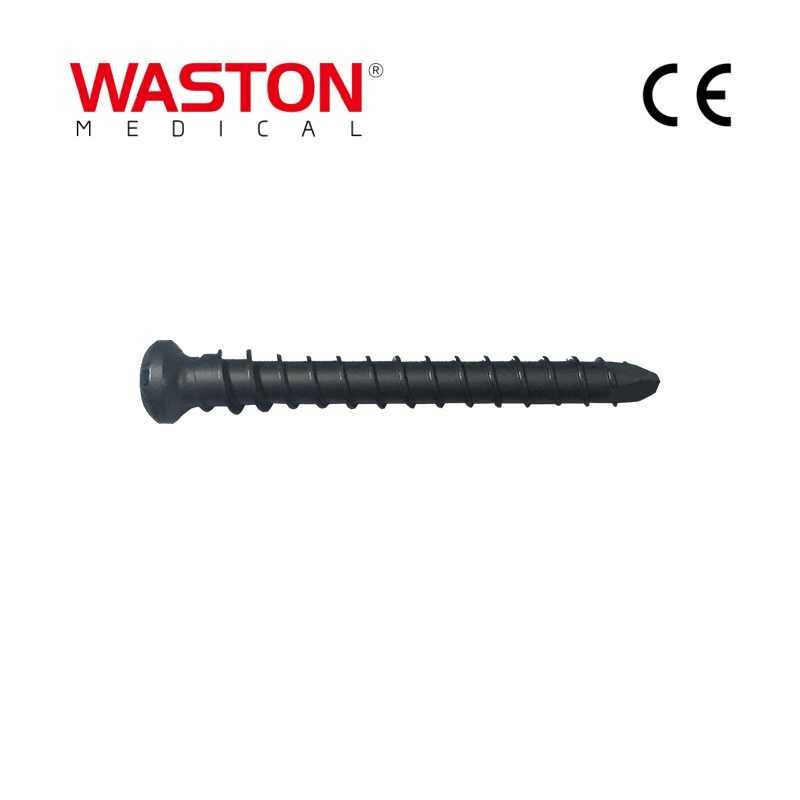
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
| Model NO. | FEMUR |
| Material | Pure Titanium / Titanium Alloy / Stainless Steel |
| Feature | Disposable |
| Certification | CE, ISO13485, SGS |
| Group | Adult |
| Package | Non-Sterile Package, Paper Plastic Roll Bag |
| HS Code | 9021100000 |

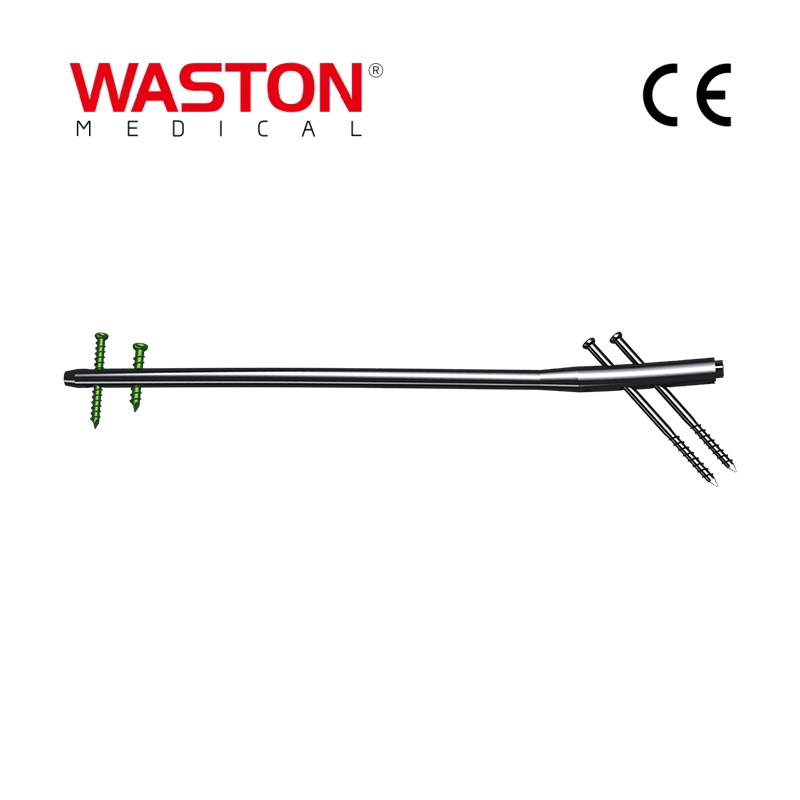
| PRODUCT CODE | L/R | HOLES | LENGTH (mm) |
|---|---|---|---|
| 10732-006 | L | 6 | 88 |
| 10732-106 | R | - | - |
| 10732-007 | L | 7 | 101 |
| 10732-107 | R | - | - |
| 10732-008 | L | 8 | 114 |
| 10732-108 | R | - | - |
| 10732-010 | L | 10 | 137 |



As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. We focus on transforming clinical experience into innovative medical solutions, pioneering global technologies such as the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plates.
Quality Assurance: We operate strictly according to the ISO13485 quality system and the CE MDD 93/42/EEC directive. Our production facility utilizes German and American-imported machining centers and Japanese-imported automatic lathes to ensure first-class quality.

Our Testing Center includes a biomechanical laboratory and a physical-chemical testing center, ensuring rigorous control over raw material procurement, product quality, and performance analysis.
Our products are exported to many countries, including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We are also a major OEM supplier to leading global players in the medical industry.



 Daher Orthopedic Implants
Daher Orthopedic Implants