
1 / 5










| Still deciding? Get samples of $ ! US$ 40/Piece Request Sample |
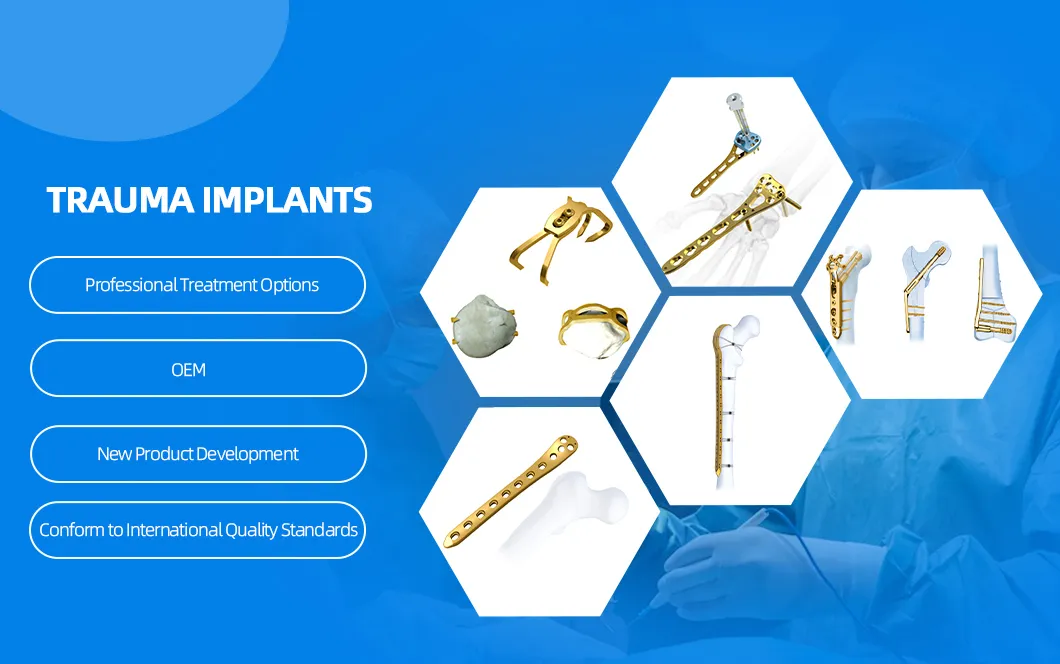
Titanium Medical Orthopedic Trauma Maxillofacial Bone Fracture Reconstruction LCP Locking Compression Plate
| Product | Holes | Direction | Material | Length (mm) |
|---|---|---|---|---|
| 2.0mm Straight Bridge Repair Skull LCP | 2 | - | Pure Titanium | 16.7 |
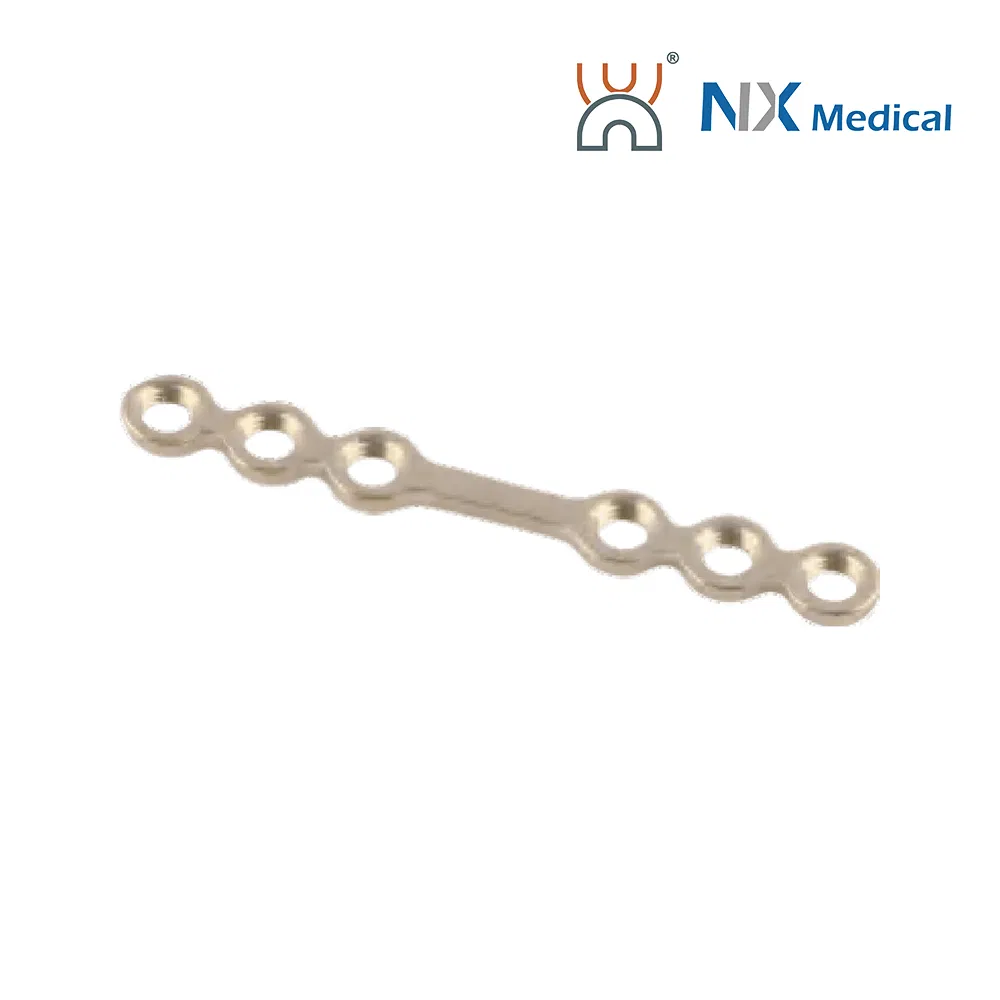
| 2.0mm Straight Bridge Repair Skull LCP | 4H, 6mm-22mm | - | Pure Titanium | 20.7 - 36.7 |
| 2.0mm Arc Skull Reparing LCP | 4-10 | - | Pure Titanium | 22.5 - 52.6 |
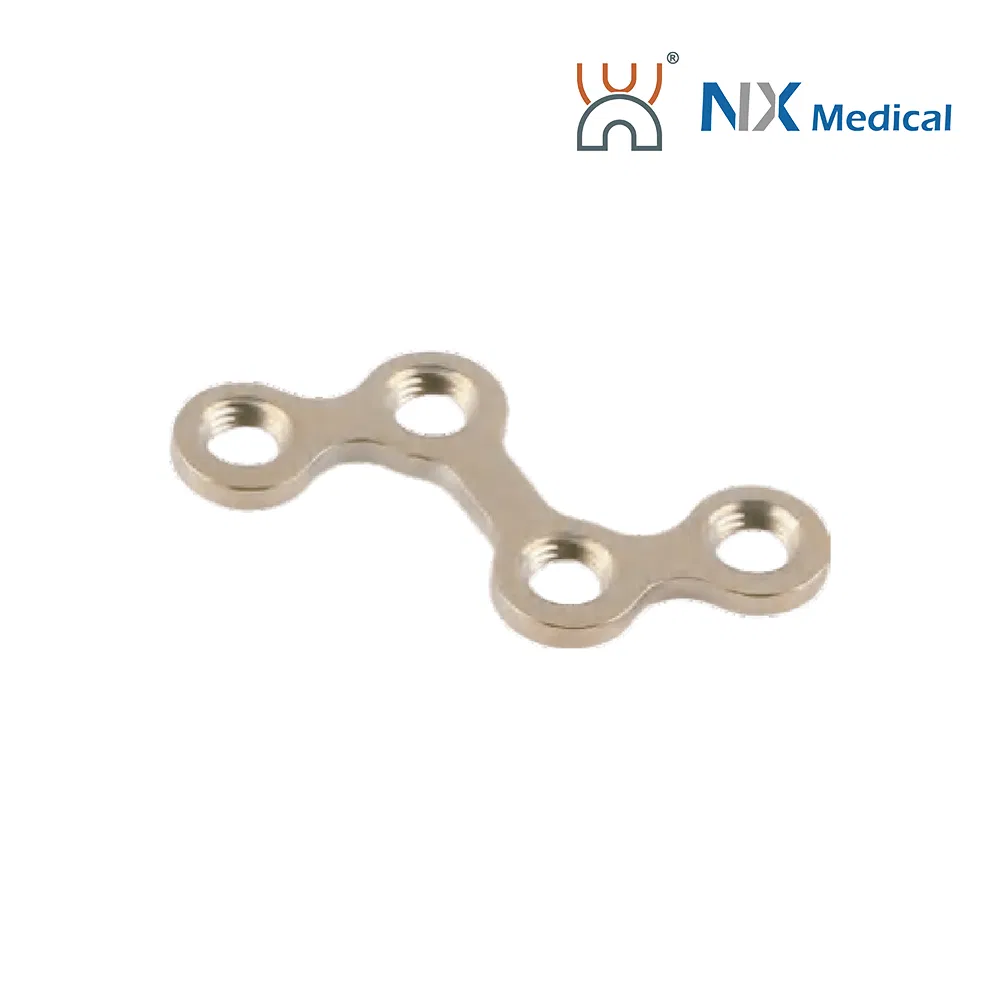
| 2.0mm T-type Cranioplasty LCP | 2+3H / 2+5H | - | Pure Titanium | 21.9 - 33.9 |
| 2.0mm Oblique T-type Cranioplasty | 2+4H | Left/Right | Pure Titanium | 29.2 |
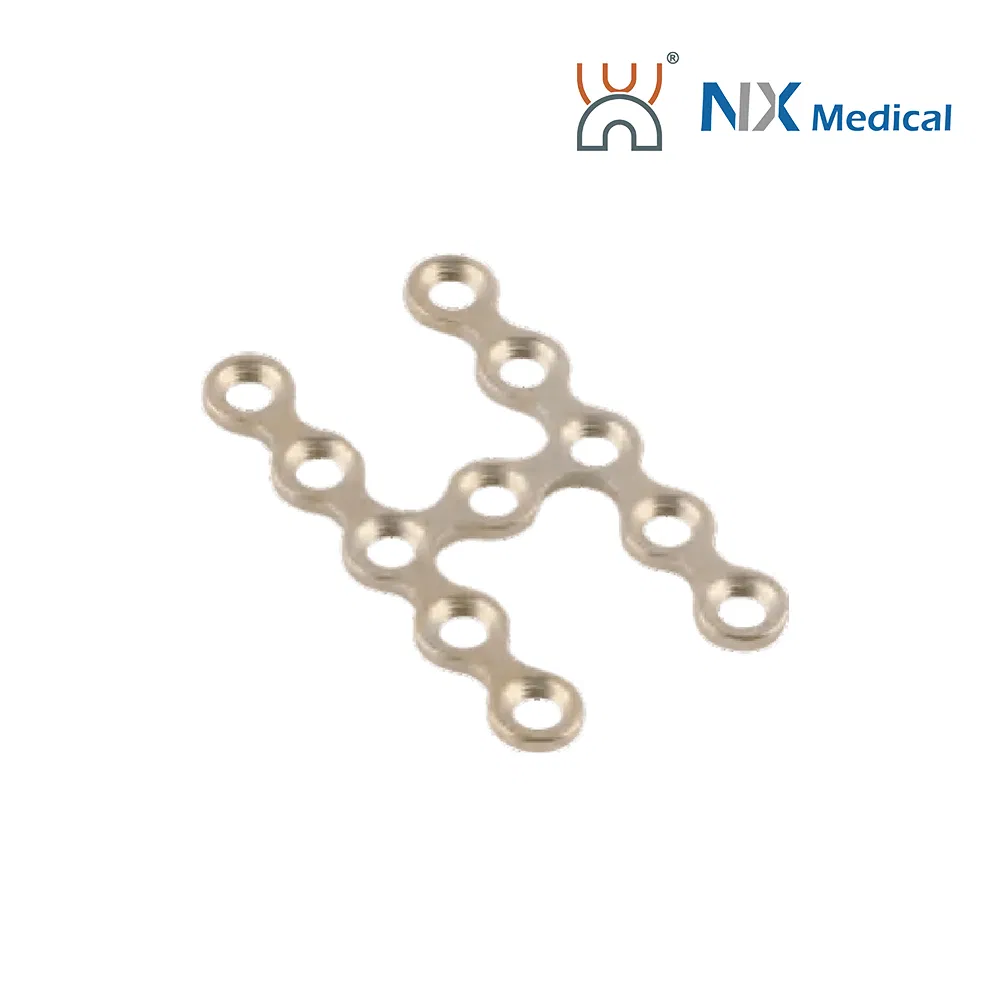
| 2.0mm X-type Cranioplasty LCP | 3+3H, 6-12mm | - | Pure Titanium | 19.5 - 25.5 |
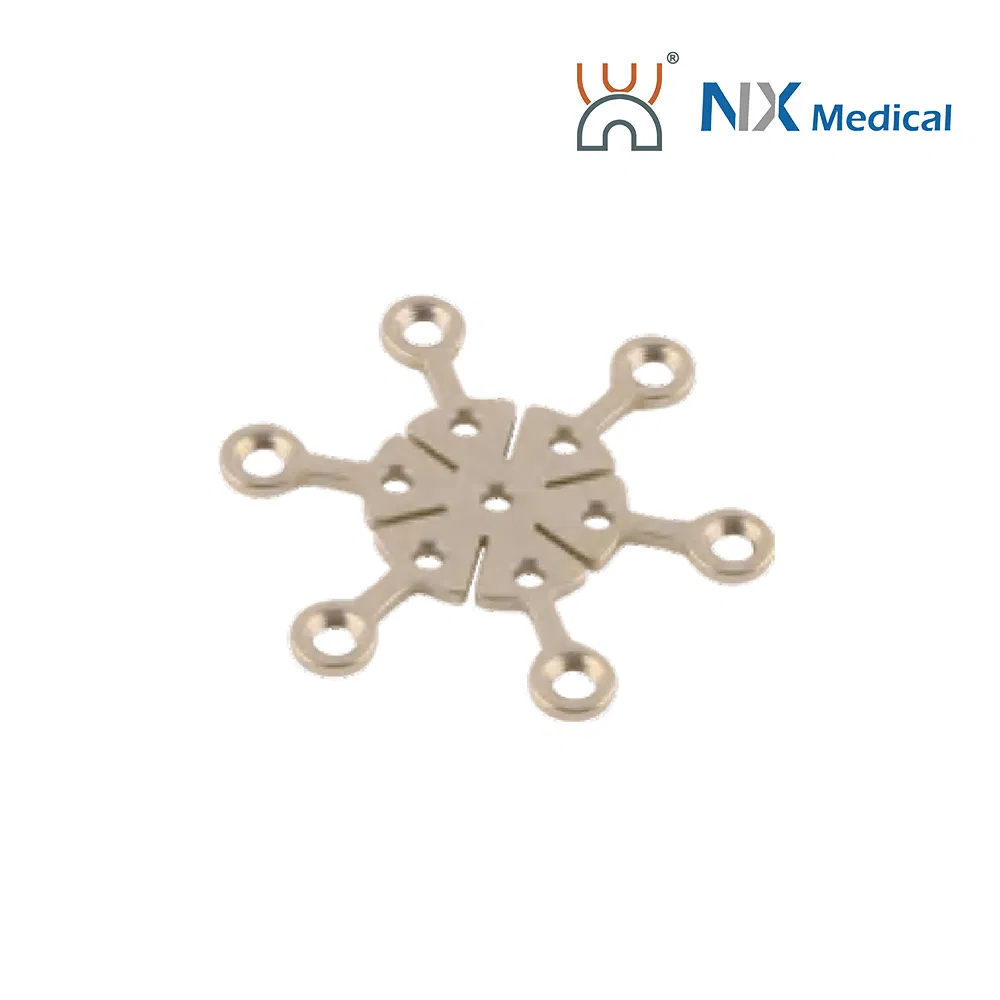
| 2.0mm H-type Nasal LCP | 6+6H | Straight/L/R | Pure Titanium | 28.7 |
| 2.0mm Z-type Cranioplasty LCP | 2+2H, 8-16mm | Left/Right | Pure Titanium | 15.8 - 23.8 |
| 2.0mm Y-type Cranioplasty LCP | 3+2H / 3+4H | - | Pure Titanium | 21.0 - 40.0 |


The manufacturer is a leading orthopedic product provider located in East China, specializing in cost-effective surgical and reliable therapeutic solutions. The comprehensive range includes orthopedic trauma implants, spinal implants, and professional surgical instruments utilized worldwide.
The trauma system focuses on locking plates, intramedullary nails, cannulated screws, and cable systems. All products are manufactured under strict internal quality systems and are certified by ISO13485, TUV CE0123, and NMPA standards.





 Daher Orthopedic Implants
Daher Orthopedic Implants