
1 / 5










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |


| Product | Size (mm) | Material |
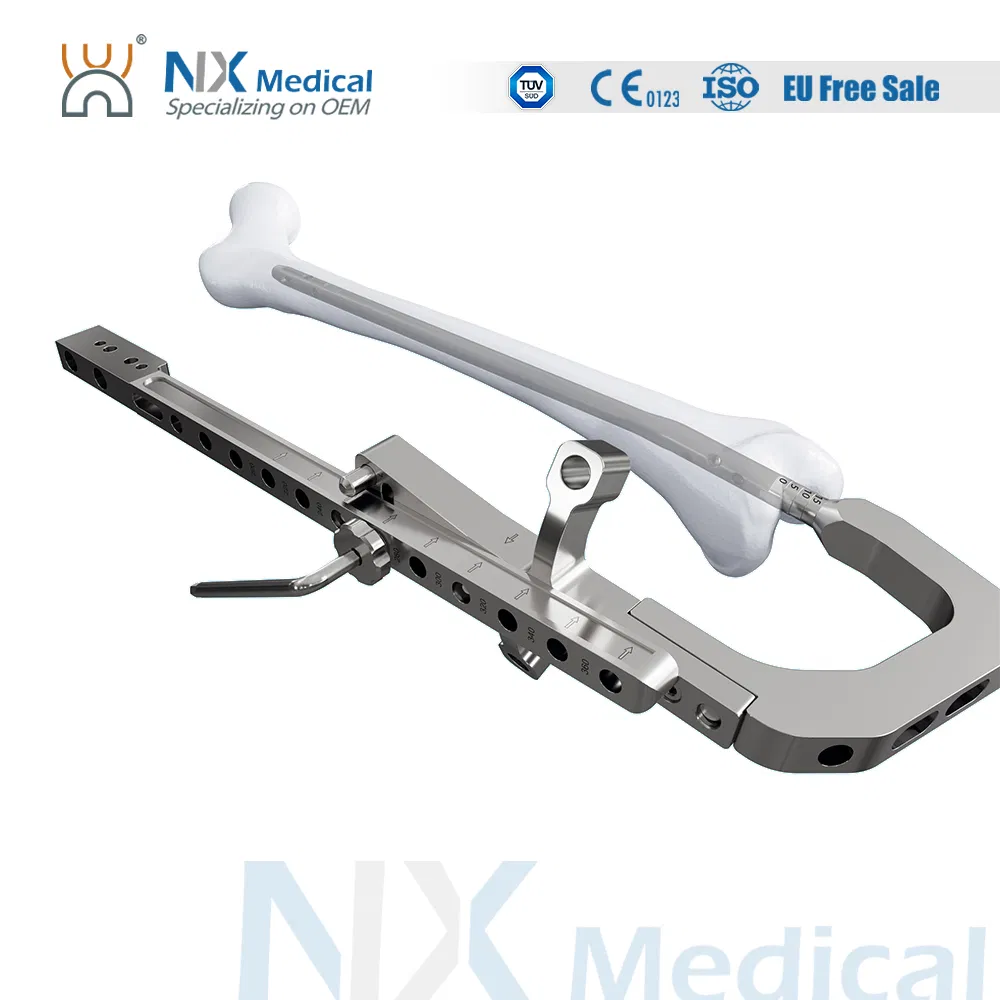
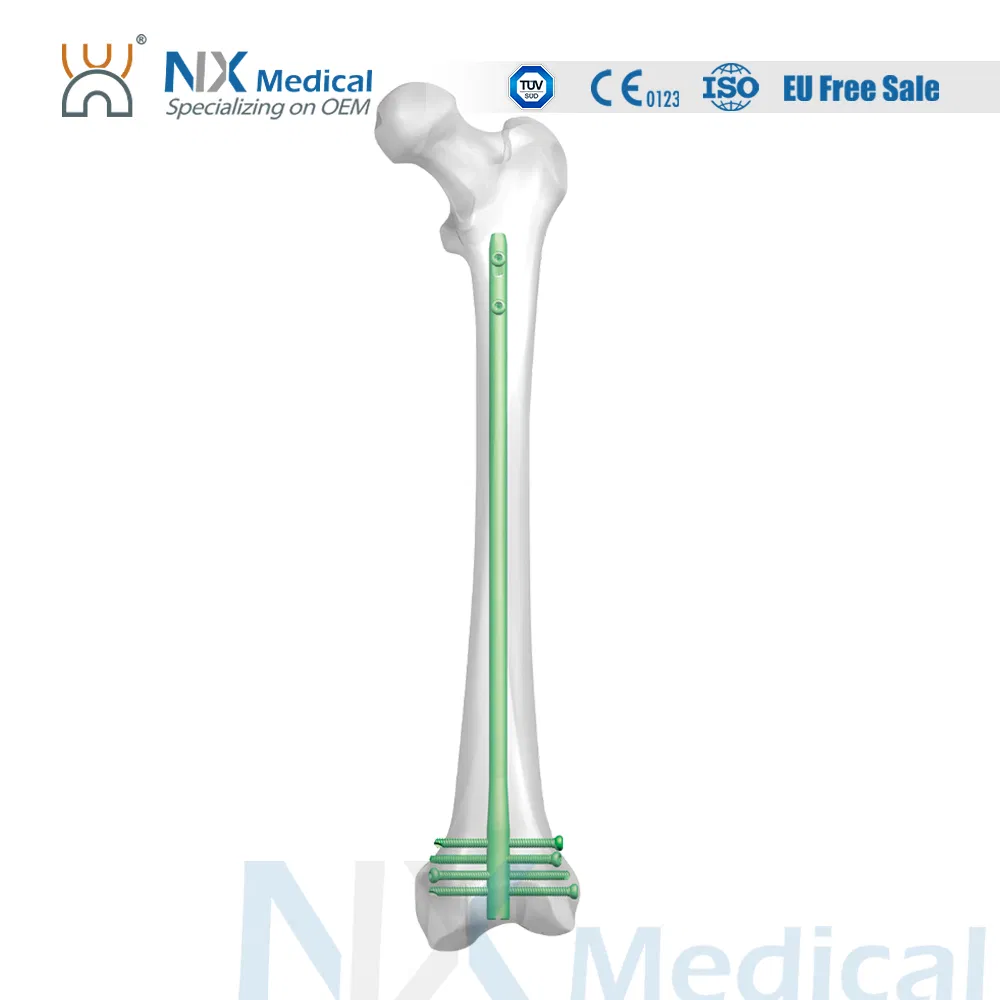
| Retrograde Femoral Nail | 9 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy (TA) |
| 10 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy (TA) | |
| 11 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy (TA) | |
| 12 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy (TA) | |
| φ4.8mm IM Nail Interlocking Screw | 26 / 28 / 30 / 32 / 34 / 36 / 38 / 40 / 42 / 44 / 46 / 48 / 50 / 52 / 54 / 56 / 58 / 60 / 64 / 68 / 72 / 76 / 80 / 84 | Titanium Alloy (TA) |
| Retrograde Femoral Nail End Cap | Extention: 0 / 5 / 10 / 15 | Titanium Alloy (TA) |
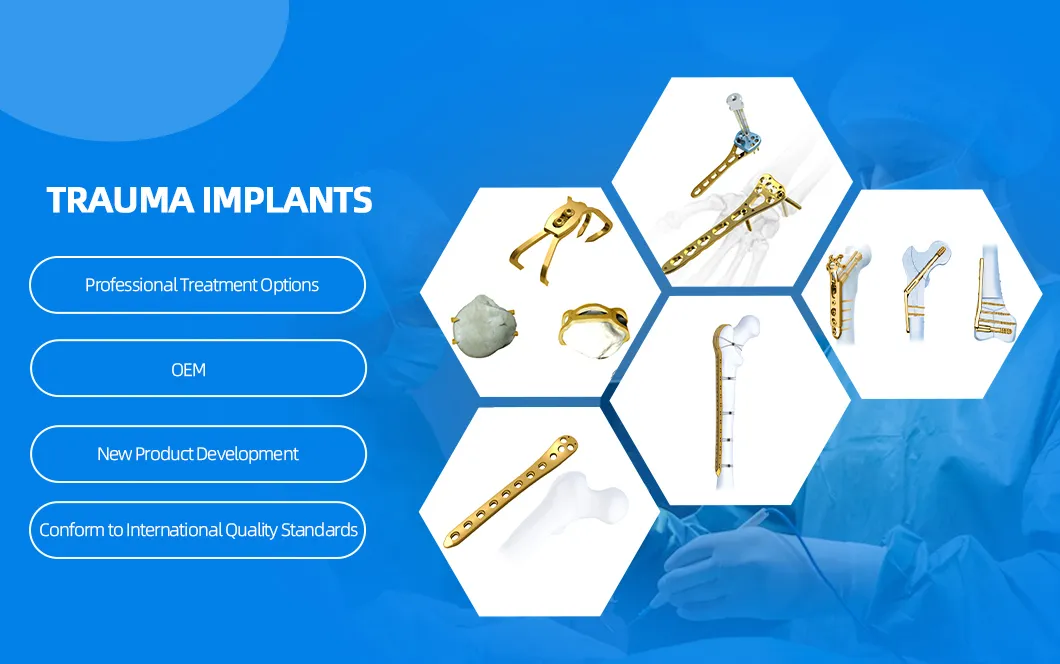
We are a leading Orthopedic product manufacturer providing a comprehensive range of cost-effective surgical and reliable therapeutic solutions to Orthopedic surgeons and patients. Our facility offers superior and various kinds of Orthopedic trauma implants, spinal implants, and surgical instruments worldwide. We are committed to the global industry through continuous investment in R&D.



Our quality system is established to fulfill frequent self-audits and complies with ISO13485. Products are certified by TUV CE0123 and NMPA. We also serve as a major OEM supplier to large global players in the orthopedic industry, ensuring the safest and most advanced implants and technologies for rapid patient recovery.


 Daher Orthopedic Implants
Daher Orthopedic Implants