














| Still deciding? Get samples of $ ! US$ 160/Piece Request Sample |
As a national high-tech enterprise, our R&D investment consistently exceeds 8% of total sales. This commitment enables the transformation of clinical expertise into innovative medical achievements. Several products hold national invention and utility patents, featuring revolutionary technology such as Rev Drill systems, Three-Row-Staplers, and specialized fixation plates.

Our operations strictly adhere to ISO13485 quality systems and CE MDD 93/42/EEC directives. By standardizing every detail of management, we maintain an international level of quality control.
Our facility is equipped with German and American-imported machining centers, along with Japanese automatic lathes. Our advanced Testing Center includes biomechanical and physical-chemical laboratories to ensure rigorous raw material procurement control and comprehensive performance analysis.

Our orthopedic solutions are exported globally, including regions such as Europe, Southeast Asia, and Latin America. We also serve as a major OEM supplier for global medical industry leaders.







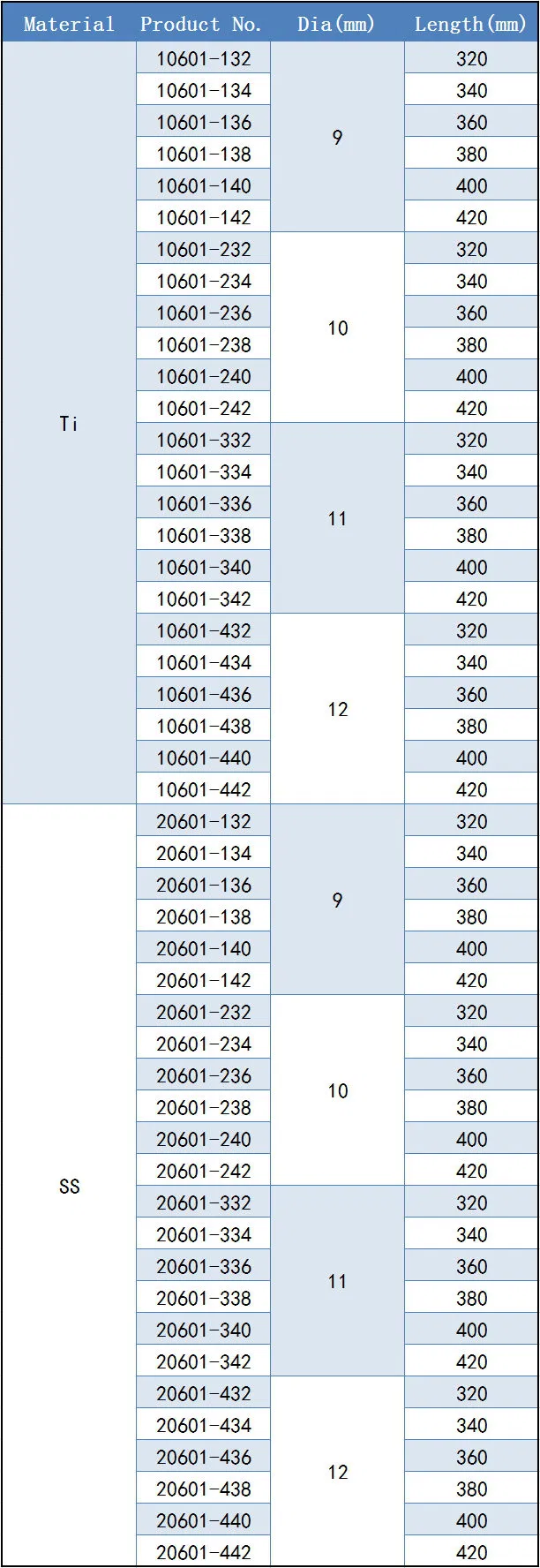
 Daher Orthopedic Implants
Daher Orthopedic Implants