Nagoya, the heart of Japan's Aichi Prefecture, stands as a global titan of industrial manufacturing and technological innovation. Known historically for its "Monozukuri" (the art of making things), Nagoya has transitioned from traditional heavy industries into a specialized hub for high-precision medical device manufacturing. As a leading Intramedullary Nails Supplier in Nagoya, we leverage this unique industrial ecosystem to produce implants that meet the world's most rigorous anatomical and mechanical requirements.
The synergy between Nagoya's aerospace engineering expertise and medical technology has led to the development of intramedullary nails with unmatched fatigue resistance and biocompatibility. In Nagoya, the medical device sector benefits from advanced CNC machining, localized titanium smelting technologies, and a workforce trained in the precision-first culture of the automotive and robotics industries. This ensures that every intramedullary nail we supply is not just a tool, but a masterpiece of surgical engineering.
The intramedullary (IM) nail market is undergoing a radical shift toward minimally invasive surgery (MIS) and patient-specific solutions. In Nagoya, we are at the forefront of these trends, integrating AI-assisted design and advanced metallurgy into our production lines. Modern trauma surgery demands implants that allow for early weight-bearing and accelerated rehabilitation—factors that are critical in Japan’s rapidly aging society.
Key Trends in Intramedullary Fixation:
As a prominent Intramedullary Nails Factory in Nagoya, we collaborate closely with university hospitals and clinical researchers to refine our nail geometries. Our expert systems, such as the Expert Humeral and Tibial proximal interlocking nails, are designed with anatomical curvatures specific to diverse patient demographics, ensuring better fit and easier insertion.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product. Our factory has passed ISO 9001 and ISO 13485 certifications, and our products exceed international quality and safety standards, including CE and FDA certifications.
In Nagoya, our quality control philosophy is simple: Quality and integrity first, striving for excellence, and pursuing the highest standards. A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential in the operating room.

Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.
 Nagoya Factory: Expert Femur Intramedullary Nail System
Nagoya Factory: Expert Femur Intramedullary Nail System
 Nagoya Grade Interlocking Nail - Multi-Color Anodization
Nagoya Grade Interlocking Nail - Multi-Color Anodization
 PFNA Proximal Femur Nail - Precision Engineered in Nagoya
PFNA Proximal Femur Nail - Precision Engineered in Nagoya
 Nagoya Retrograde Intramedullary Femur Interlocking Nail
Nagoya Retrograde Intramedullary Femur Interlocking Nail
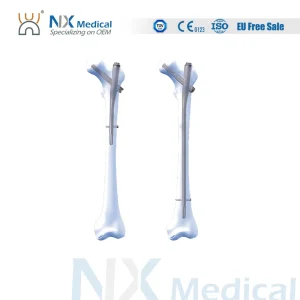 NX-Series: Nagoya Proximal Femoral Interlocking Nail
NX-Series: Nagoya Proximal Femoral Interlocking Nail
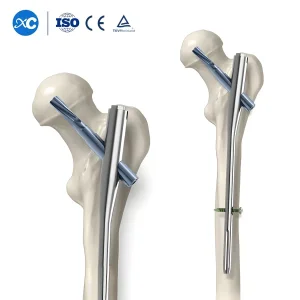 Nagoya Trauma Surgery: Gama Interlocking PFNA Nail
Nagoya Trauma Surgery: Gama Interlocking PFNA Nail
 Nagoya Premium Expert Tibial Proximal Interlocking Nail
Nagoya Premium Expert Tibial Proximal Interlocking Nail
 Nagoya Humeral Proximal Interlocking Nail - Blade Locking
Nagoya Humeral Proximal Interlocking Nail - Blade Locking
Nagoya is home to some of Japan's most prestigious medical institutions, such as Nagoya University Hospital and Nagoya City University Hospital. These centers are pioneers in utilizing intramedullary nails for complex trauma and geriatric fractures. Our products are specifically optimized for the Japanese surgical environment, where the emphasis is on precision, minimal incision, and patient safety.
In cases of high-energy trauma often seen in Nagoya’s urban transport networks, or osteoporotic fractures in the elder population, our Titanium PFNA Nails and Multi-lock Humeral Nails provide the stable internal fixation required for rapid recovery. The use of titanium alloys is particularly favored in the Nagoya market due to their excellent strength-to-weight ratio and MRI compatibility, which is essential for the high-tech diagnostic protocols standard in Japanese hospitals.
Furthermore, our Suprapatellar Tibial Nails have seen increased adoption in Nagoya-based clinics. This approach allows surgeons to perform the procedure with the knee in a semi-extended position, providing better radiographic visualization and potentially reducing chronic anterior knee pain—a common complication in traditional tibial nailing.
Whether you are a hospital administrator, a clinical distributor, or a surgical specialist, our Nagoya-standard intramedullary systems offer the reliability and innovation you need.