Precision-engineered implants for total joint restoration
The global Ankle Joint Replacement (AJR) market, also known as Total Ankle Arthroplasty (TAA), is undergoing a monumental shift. Traditionally, ankle fusion (arthrodesis) was the "gold standard" for treating end-stage ankle arthritis. However, the industrial landscape has evolved. Modern medical engineering now prioritizes mobility preservation, leading to a surge in demand for high-tech prosthetic solutions. As a leading manufacturer and supplier, we are at the forefront of this transition, providing implants that mimic the complex kinematics of the human ankle.
Today, the market is driven by an aging population, an increase in sports-related injuries, and significant advancements in material sciences such as Titanium-6Al-4V ELI and Highly Cross-linked Polyethylene (HXLPE). Our role as a top-tier supplier involves bridging the gap between clinical needs and industrial manufacturing capabilities, ensuring that surgeons in Europe, North America, and emerging markets have access to long-lasting, biocompatible joint systems.
The Future of Orthopedic Implant Manufacturing
The industry is moving toward Patient-Specific Implants (PSI). Additive manufacturing allows us to create porous structures that encourage better osseointegration, significantly reducing the risk of implant loosening.
Artificial Intelligence is now used to analyze preoperative CT scans, allowing for the precise placement of ankle components. Our instrument sets are designed to integrate seamlessly with these digital workflows.
From PEEK (Polyetheretherketone) to advanced ceramic coatings, the focus is on reducing wear rates and increasing the lifespan of the prosthesis to 15-20 years or more.
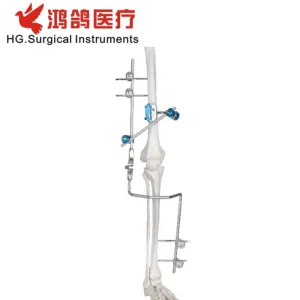
For complex cases where total replacement is not immediately viable, our Orthopedic Instrument Combination External Fixator provides the stability required for ankle fusion and joint reconstruction. This system is a testament to our manufacturing precision, utilizing medical-grade alloys that offer high strength-to-weight ratios.
Designed for multi-planar stability, it allows for micromotion which is essential for bone healing while maintaining rigid alignment. As a global supplier, we provide these systems to trauma centers and orthopedic hospitals worldwide.
International procurement managers prioritize three pillars: Quality, Cost, and Lead Time. In the competitive landscape of ankle joint replacement manufacturing, being a supplier means more than just providing a product; it means providing a localized solution. For instance, in the European market, there is a high demand for cementless fixation due to the active lifestyle of the patient base. Conversely, in Southeast Asia, cost-effectiveness and comprehensive instrument sets are vital for expanding healthcare access.
Our factory addresses these needs by offering modular designs. We understand that a "one size fits all" approach doesn't work for global orthopedics. Whether it's post-traumatic arthritis in a 40-year-old athlete or rheumatoid arthritis in a 70-year-old patient, our diverse catalog—ranging from total ankle replacements to hindfoot fusion nails—ensures every surgical scenario is covered.
Furthermore, we support OEM and ODM services. Many global brands look to Chinese factories for high-precision manufacturing under their own labels. Our ISO 13485 certified facility provides the clean-room environment and CNC machining accuracy required to meet the most stringent FDA and CE regulations.
The Hindfoot Fusion Ankle Intramedullary Nail is a pinnacle of our titanium interlocking technology. Engineered for hospital surgical departments that demand excellence, this nail provides superior compression and rotational stability.
By utilizing advanced titanium alloys, we ensure the implant is both lightweight and incredibly durable. The interlocking mechanism is designed for ease of use, reducing time in the operating room and improving patient outcomes in complex ankle surgeries.

Combining Scale, Speed, and Unmatched Quality
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product. Our strategic location in China's industrial hub allows us to leverage a massive supply chain, ensuring that we can offer competitive pricing without compromising on the medical-grade quality that patients' lives depend on.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards. Every ankle prosthesis and instrument set that leaves our facility undergoes a rigorous 12-point inspection, including fatigue testing, biocompatibility verification, and dimensional accuracy checks using laser-guided measurement systems.

A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential. This collaborative approach allows us to iterate designs quickly based on real-world surgical feedback. For example, our Knee and Ankle Instrument Sets have been refined over dozens of versions to ensure maximum ergonomic efficiency for the surgeon.
Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia, making us a trusted partner in the global orthopedic community.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.
Full-range solutions for Hip, Knee, and Ankle Reconstruction