Innovative Intramedullary Nailing Systems for Complex Fracture Fixation
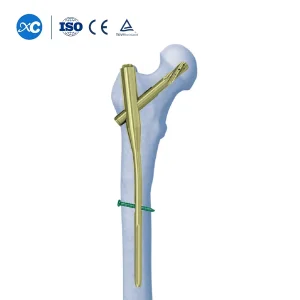
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
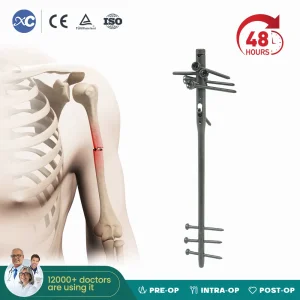
Titanium Orthopedic Surgical Implant Suprapatellar Approach Tibial Intramedullary Nail

Canwell Tibial Nail Intramedullary Nail Interlocking Cannualted Nail Canetn Gamma Nail


Orthopedic Implant Elastic Intramedullary Nail, Orthopedic Implant Ulna Radius Intramedullary Nail
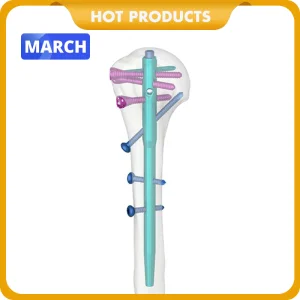

The global orthopedic trauma market has witnessed a seismic shift in the last decade, with Tibial Intramedullary (IM) Nailing emerging as the "Gold Standard" for the treatment of tibial shaft fractures. As populations age and urbanization leads to a higher incidence of high-energy road traffic accidents, the demand for efficient, reliable, and minimally invasive fixation methods has skyrocketed.
In the current industrial context, manufacturers are no longer just suppliers; they are critical partners in the surgical workflow. The integration of biocompatible Titanium alloys (Grade 5) and Stainless Steel 316L has allowed for implants that offer superior fatigue resistance while promoting early weight-bearing and faster patient recovery. Globally, the market is characterized by a push toward decentralized manufacturing, where OEM/ODM factories in China provide the technological backbone for international brands across Europe, North America, and the Middle East.
The Tibial Nail segment accounts for over 35% of the global long-bone fracture market, driven by its clinical efficacy in both simple and comminuted fractures.
Focus is shifting toward "Smart Nails" and anatomically pre-contoured designs that minimize the need for intra-operative bending.
The intramedullary approach preserves the periosteal blood supply, a critical factor for accelerated bone healing in modern trauma care.
The future of tibial nailing is defined by Precision and Digitalization. We are observing several key trends that are reshaping how OEM/ODM Tibial Intramedullary Nail Manufacturers operate:
Success Rate in Shaft Fractures
Reduction in Surgery Time
Years Fatigue Life
Global procurement teams in the medical sector face the dual challenge of cost containment and uncompromising quality. Whether it’s a government tender in Southeast Asia or a private hospital group in South America, the requirement is clear: FDA/CE-certified products that offer the same mechanical performance as "Big Tech" brands but at a sustainable price point.
Localized Application Scenarios:
As a premier China-based OEM/ODM Tibial Intramedullary Nail Manufacturer, we bridge the gap between high-end engineering and manufacturing efficiency. Our advantage is built on four pillars:
We utilize Swiss-type lathes and 5-axis machining centers to ensure tolerances within microns, ensuring every interlocking screw fits perfectly.
Our ISO 13485 certified facility implements 100% inspection protocols, including fatigue testing and chemical composition analysis of raw materials.
From concept drawings to sterile-packaged finished products, we provide a full-stack solution for brands looking to expand their trauma portfolio.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product. Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential. Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.

Explore our wide range of interlocking and intramedullary nailing systems

China Factory Nx Medical Orthopedic Trauma Implant Titanium Retrograde Tibial Intramedullary Nail
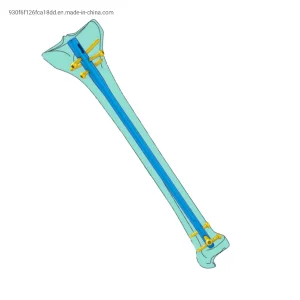
Intramedullary Nail System Expert Tibial Interlocking Nail Suprapatellar Approach

Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
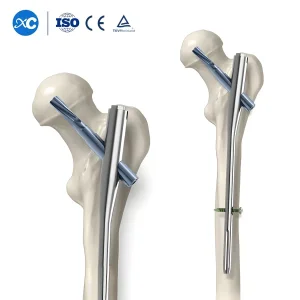
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail

Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail

High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors

High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
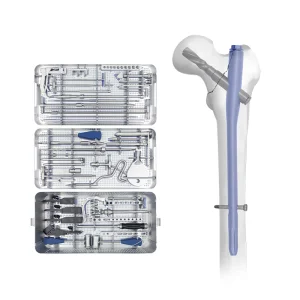
Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set