The global orthopedic market is undergoing a significant paradigm shift. For decades, Total Knee Arthroplasty (TKA) was the gold standard for late-stage osteoarthritis. However, as medical diagnostics improve and the demand for "joint-preserving" surgeries rises, Unicompartmental Knee Arthroplasty (UKA), also known as partial knee replacement, has emerged as a preferred solution for patients with single-compartment damage. Today, the global UKA market is expanding at a CAGR of approximately 5-7%, driven by an aging population in Europe and North America and a burgeoning middle class in Asia-Pacific seeking high-quality healthcare.
As a leading Custom OEM Unicompartmental Knee Arthroplasty factory, we recognize that the industrial status of this sector is no longer just about manufacturing; it's about the convergence of biotechnology, material science, and robotic-assisted surgery. Modern factories must now integrate high-precision CNC machining with AI-driven design protocols to meet the stringent demands of global healthcare providers.
The rise of Mako-like robotic systems requires implants with extreme tolerances. Our OEM manufacturing adapts to these robotic ecosystems seamlessly.
Transitioning from standard Cobalt-Chrome to advanced Oxinium and highly cross-linked polyethylene (UHMWPE) to reduce wear and extend implant life.
Utilizing AI to analyze CT scans for Patient-Specific Instrumentation (PSI), ensuring that every partial knee replacement is anatomically perfect.
Our factory specializes in the production of high-grade femoral components and tibial trays. By using advanced investment casting and vacuum heat treatment, we ensure that every piece of the Custom OEM Unicompartmental Knee Arthroplasty system meets the mechanical stresses required for active patients.
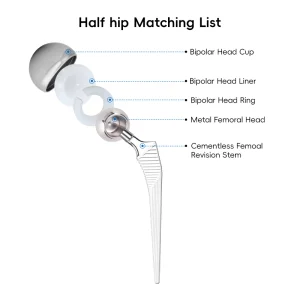
The interface between the implant and the bone is critical. We offer both cemented and cementless options, utilizing porous titanium coatings that encourage rapid osseointegration.
Global medical device distributors and hospital procurement groups are facing unprecedented pressure. On one hand, there is a push for cost containment within public health systems (like the NHS in the UK or the GPO systems in the USA). On the other hand, there is no room for compromise on clinical outcomes. This has led to a massive surge in demand for reliable OEM partners from China who can offer "Western quality at Eastern costs."
Procurement officers are now looking beyond just the price tag. They require partners with robust quality management systems (ISO 13485), clear regulatory pathways (CE marking, FDA 510k), and the ability to scale production during supply chain disruptions. Our factory has positioned itself as a strategic node in this global supply chain, offering end-to-end solutions from design to sterile packaging.
Furthermore, the demand for "Private Label" orthopedic implants is growing. We empower local brands in South America, Africa, and Southeast Asia by providing them with high-end UKA systems that they can market under their own identity, backed by our manufacturing excellence.
A successful UKA procedure is as much about the tools as it is about the implant. We provide comprehensive, ergonomic instrument sets designed to minimize surgical time and improve accuracy. Our kits are compatible with minimally invasive surgery (MIS) techniques, reducing patient recovery time.
From precision saws to tensioners, every tool is crafted from medical-grade stainless steel, ensuring durability through thousands of sterilization cycles.
Ambulatory Surgery Centers (ASCs) are the fastest-growing segment for UKA in the US. These centers require high-efficiency, "single-use" instrument options and implants that allow for same-day discharge. Our OEM services include developing specialized packaging for this "fast-track" surgery model.
In countries like India, Vietnam, and Indonesia, the focus is on durability and affordability. Our UKA solutions are designed to be "user-friendly" for surgeons who may be transitioning from TKA, providing them with reliable, repeatable outcomes in diverse clinical settings.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.

When discussing Custom OEM Unicompartmental Knee Arthroplasty Factories & Exporters, one must delve into the mechanical engineering that makes these implants successful. A UKA system typically consists of three components: a metallic femoral component, a metallic tibial tray, and a plastic (UHMWPE) insert. The challenge in OEM manufacturing lies in the thinness of the components. Unlike TKA, UKA requires removing very little bone, meaning the implants must be thin yet incredibly strong to withstand the weight of the patient.
Our production line utilizes 5-axis simultaneous CNC milling machines that can achieve tolerances as low as 5 microns. This precision is essential for the "mobile-bearing" UKA designs, where the plastic insert moves freely on the tibial tray, mimicking the natural kinematics of the human knee. By reducing the friction coefficient through mirror-polishing techniques, we significantly lower the risk of aseptic loosening, the primary cause of UKA failure.
The next decade of arthroplasty will be defined by "Smart Implants." As a forward-thinking factory, we are already investigating the integration of micro-sensors into the tibial tray to monitor post-operative load distribution and early infection markers. While still in the R&D phase, our commitment to Custom OEM Unicompartmental Knee Arthroplasty means we are preparing our infrastructure to lead this revolution. We believe that by combining traditional craftsmanship with AI-driven analytics, we can provide surgeons with tools that don't just replace a joint, but restore a lifestyle.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers. Whether you are a large-scale distributor in Europe or a specialized medical group in South America, our factory is equipped to handle your Custom OEM Unicompartmental Knee Arthroplasty needs with precision, speed, and integrity.