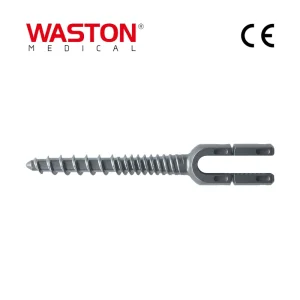
 Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Bone Graft Implant M9 Spinal Fixation System
Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Bone Graft Implant M9 Spinal Fixation System
 Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Bone Graft Implant O-Max Cage System
Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Bone Graft Implant O-Max Cage System
 Sports Medicine Interference Screws for Acl Reconstruction Peek and Cannulated Screws
Sports Medicine Interference Screws for Acl Reconstruction Peek and Cannulated Screws
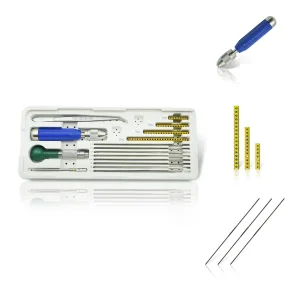 Veterinary Fixator Mini External Fixation System for Exotic Pet Surgery
Veterinary Fixator Mini External Fixation System for Exotic Pet Surgery
 Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
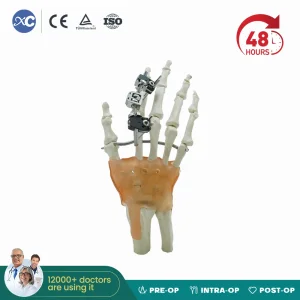 High Quality Mini Fragment External Fixator Orthopedic Medical Metacarpals External Fixation
High Quality Mini Fragment External Fixator Orthopedic Medical Metacarpals External Fixation
 Maxillofacial Locking Micro/Mini & Reconstruction Implant
Maxillofacial Locking Micro/Mini & Reconstruction Implant
 Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
The global Graft Fixation Devices market is undergoing a radical transformation. As the global population ages and sports-related injuries (such as ACL and PCL tears) increase, the demand for reliable, high-performance orthopedic implants has reached an all-time high. Currently, the market is valued in the billions, driven by advancements in material science—moving from traditional stainless steel to medical-grade titanium and biocompatible PEEK (Polyether ether ketone).
Major healthcare providers in North America, Europe, and Asia-Pacific are increasingly seeking "Value-Based Healthcare" solutions. This means they require devices that not only offer clinical excellence but also cost-efficiency. This is where China's best graft fixation factories play a pivotal role, bridging the gap between high-end innovation and large-scale manufacturing affordability.
Stay ahead with the latest technological shifts in the surgical instrument industry.
The industry is shifting toward materials that the body can naturally absorb over time, eliminating the need for secondary removal surgeries.
Customized 3D-printed titanium implants allow for patient-specific geometry, ensuring a perfect fit and faster osseointegration.
Integration of IoT sensors in orthopedic instruments to monitor bone healing and stress distribution post-surgery is the next frontier.

When sourcing Graft Fixation Devices, global procurement officers look for the "Triple Play": Quality, Speed, and Cost. Our facility in China excels in all three:
Our products are engineered for diverse surgical environments across the globe.
Our Interference Screws and ACL/PCL kits are the preferred choice for outpatient clinics in Europe and the Middle East, where rapid recovery is the priority for athletes.
In high-traffic urban hospitals, our Variable Angle Distal Volar Radius Locking Plates provide the versatility needed to treat complex bone fractures quickly and effectively.
Exotic pet surgery requires precision on a miniature scale. Our mini external fixation systems are specially calibrated for small-animal orthopedic procedures.
The M9 and 11412 Fixation Rod series are designed for advanced spinal stabilization, supporting long-term recovery for patients with chronic degenerative conditions.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential.
Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.
 Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant 11412 Fixation Rod IV 6.0 Spinal Fixation System
Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant 11412 Fixation Rod IV 6.0 Spinal Fixation System
 Orthopedic Spinal Fixation Medical Instrument Spine Vertebral Hook
Orthopedic Spinal Fixation Medical Instrument Spine Vertebral Hook
 Orthopedic Spinal Fixation Vertebral Hook Medical Instrument
Orthopedic Spinal Fixation Vertebral Hook Medical Instrument
 Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation Bone Fracture Implants Medical Titanium Orthopedic Surgical Plate
Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation Bone Fracture Implants Medical Titanium Orthopedic Surgical Plate
 Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation Bone Fracture Implants Medical Titanium Orthopedic Surgical Plate
Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation Bone Fracture Implants Medical Titanium Orthopedic Surgical Plate
 Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery
Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery
 Orthopedic Instrument Set for Precision Bone Fixation and Surgery
Orthopedic Instrument Set for Precision Bone Fixation and Surgery
 Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit
Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit
Procurement teams are no longer just looking for "suppliers"—they are looking for "long-term strategic partners." In the post-pandemic era, resilience in the supply chain is critical. Our factory provides this resilience through vertically integrated manufacturing. From the raw titanium ingot to the final sterilized packaging, every step is controlled in-house. This ensures that even during global logistical disruptions, our clients in Europe and America receive their orders on schedule.
Furthermore, the digital transformation of surgery means that fixation devices must be compatible with robotic-assisted surgery and navigation systems. Our latest R&D efforts are focused on ensuring that our screws and plates have the precise tolerances required for the next generation of digital operating rooms.
A graft fixation device is only as good as its clinical outcome. Whether it's the 6.0mm Pedicle Screw system or the Maxillofacial Locking Micro Implants, our design focus is on "Osseointegration"—the direct functional and structural connection between living bone and the surface of a load-bearing artificial implant. By utilizing advanced surface treatments like acid-etching and sand-blasting, we create a topography that encourages bone cells to attach and grow, significantly reducing the risk of implant rejection or loosening.