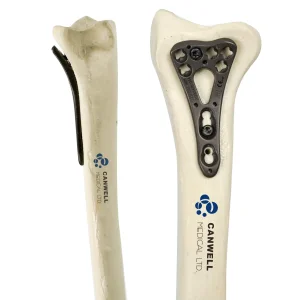
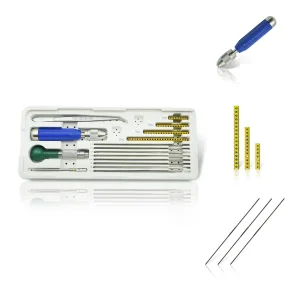
Cannulated screws have revolutionized modern orthopedic surgery, specifically in the realm of Minimally Invasive Surgery (MIS). As China's leading manufacturers and exporters of orthopedic implants, we have witnessed and driven the evolution of this technology. These screws, characterized by their hollow center designed for guide wire placement, allow surgeons to achieve precise reduction and fixation with minimal soft tissue disruption. This not only reduces operative time but also significantly enhances patient recovery rates, lowers infection risks, and minimizes postoperative pain.
In the contemporary medical landscape, the demand for high-quality cannulated screws has surged globally. From femoral neck fractures to scaphoid repairs and pelvic fixations, the versatility of these implants is paramount. Our commitment to research, development, production, and sales ensures that we provide the medical community with instruments that meet the highest international standards of efficacy and safety.







The global orthopedics market is witnessing an unprecedented transformation. As the population ages, the prevalence of orthopedic trauma and degenerative diseases has increased, driving a sustained demand for high-grade implants. Industry reports highlight that the cannulated screw segment is particularly robust, driven by the shift towards outpatient surgical procedures.
Key Trends include:
Global procurement demands are shifting. Hospitals and medical distributors are no longer just looking for the lowest price; they are seeking Total Cost of Ownership (TCO) value, which includes superior sterilization compatibility, instrument longevity, and, most importantly, stringent regulatory compliance such as CE and FDA approvals.
As a leading Chinese factory, our competitive edge lies in the synthesis of advanced manufacturing infrastructure and a culture of continuous improvement. We do not just manufacture parts; we craft life-enhancing solutions.
Our quality control philosophy is built on the pillars of Integrity, Precision, and Efficacy. Our facility has achieved ISO 9001 and ISO 13485 certifications, underscoring our commitment to quality management systems specific to medical devices. Our products are exported to over 70 countries and regions in Europe, America, Africa, and Southeast Asia, a testament to our global reliability.
We work in close collaboration with leading hospital experts, facilitating long-term technical exchanges. This ensures that our R&D team stays ahead of surgical needs, allowing us to pivot quickly and innovate based on real-world medical feedback.


Oxidation All-in-one Machine

CNC Carving Machine

Ultrasonic Cleaning Dryer

Vacuum Tempering Furnace

Digital Controlled Lathe

Leakage Current Tester

Earth Resistance Tester

Electromagnetic Dynamic Mechanics Test System