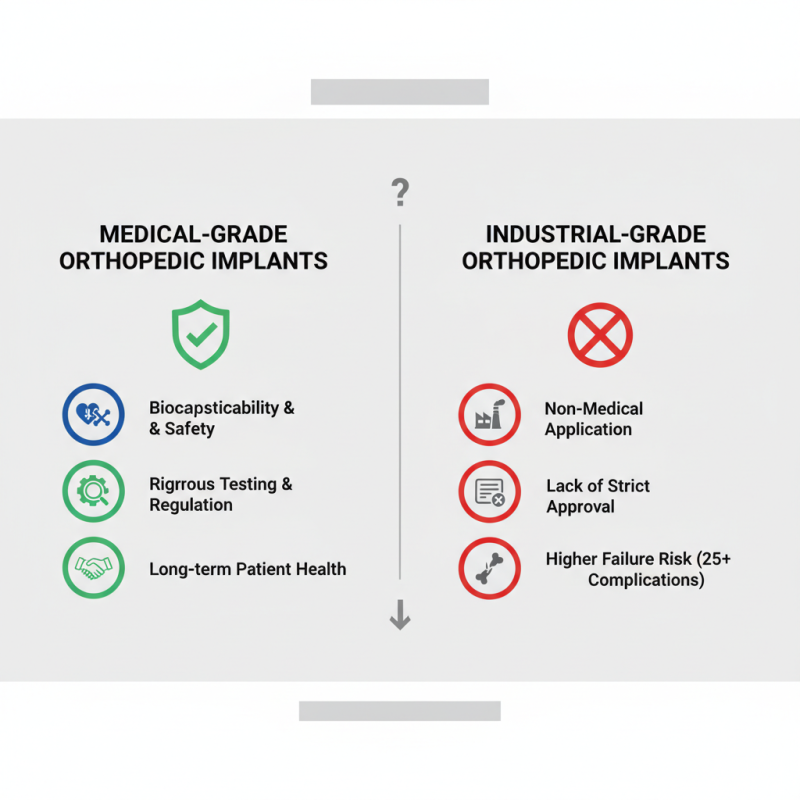
In recent years, the orthopedic implant industry has witnessed significant advancements. The distinction between medical-grade and industrial-grade implants has become a hot topic. According to a study by the Orthopedic Research Society, medical-grade implants ensure biocompatibility and safety, critical for patient health.
Experts like Dr. Emily Chen, a leading researcher in orthopedics, emphasizes, "What is the difference between medical-grade and industrial-grade orthopedic implants? It's all about quality and application." Medical-grade implants undergo rigorous testing and regulatory approval, which industrial-grade items typically do not. This creates critical safety implications.
Data indicates that failure rates for inferior-grade materials can rise significantly. In 2022, the FDA reported that over 25% of complications stemmed from non-medical-grade implants. Such statistics underline the importance of using appropriate materials. Choosing the right implant isn't just a technical decision; it's a matter of life and longevity. There is much room for improvement in education and awareness about these differences in both medical professionals and patients.
Medical grade orthopedic implants are designed with strict safety standards in mind. They are made from biocompatible materials. This means they can safely integrate with the human body. Patient safety often drives the extensive testing of these implants. Rigorous clinical trials ensure they perform well under physiological conditions. These implants are durable but also flexible enough to accommodate body movements.
Additionally, medical grade implants must resist corrosion and wear. This is vital for the longevity of an implant inside the body. In contrast, industrial grade implants may not meet the same stringent safety requirements. They could be more susceptible to failure due to environmental factors. The materials used in medical grade implants provide better performance in various conditions. Factors like blood compatibility and the body's reaction to materials are crucial considerations.
There are still questions about the long-term performance of some implants. Research continues into how different materials interact over time. Understanding these nuances can lead to significant improvements. Emphasis on detail and patient experience should guide implant design. The difference it makes in patients' lives is immeasurable, yet more insights are necessary.
When discussing orthopedic implants, materials play a crucial role in their effectiveness. Medical-grade implants are often made from high-quality titanium or cobalt-chromium alloys. These metals provide excellent biocompatibility, meaning they work well with the human body. They resist corrosion and wear, ensuring long-lasting performance in surgical settings.
On the other hand, industrial-grade implants may use more affordable materials. Common choices include stainless steel or lower-grade titanium. While these materials can still fulfill their intended purpose, they often lack the necessary quality for medical applications. This can lead to issues like increased wear or allergic reactions in some patients.
Moreover, the manufacturing process for medical-grade implants is usually stricter, with tight quality controls in place. This ensures that every component meets health regulations. Conversely, industrial-grade implants might not go through the same rigorous testing. This difference raises concerns about their long-term reliability in the body. The choice of materials can significantly impact patient outcomes and should be carefully considered.
Medical grade implants are held to stringent regulatory standards, ensuring safety and efficacy for patients. These standards are enforced by agencies responsible for public health. For instance, the testing process involves rigorous biocompatibility assessments. All components must be thoroughly examined for potential reactions in the human body. This helps to minimize risks associated with allergic responses or infections.
In contrast, industrial grade implants do not follow the same level of scrutiny. These products may comply with general manufacturing standards, but they lack specific oversight for human use. The materials might be less tested for long-term effects in medical applications. As a result, there are concerns regarding durability and patient safety.
The gap between the regulatory standards for medical grade and industrial grade implants raises questions about safety. Many might assume that all implants are equally reliable. However, the differences in quality control and testing phases can have significant implications. This discrepancy underlines the importance of understanding these categories before making informed decisions about orthopedic solutions.
| Criteria | Medical Grade Implants | Industrial Grade Implants |
|---|---|---|
| Regulatory Standards | Must comply with FDA and ISO regulations | Applicable industrial standards, less stringent |
| Material Certification | Requires biocompatibility testing | Material properties but no biocompatibility |
| Sterilization Requirements | Must be sterilized to prevent infection | Sterilization is not mandatory |
| Usage Duration | Designed for long-term implantation | Primarily for short-term use |
| Traceability and Documentation | Extensive documentation and traceability | Minimal documentation required |
Biocompatibility is crucial in orthopedic implants. Medical-grade implants are designed for the human body. They must not trigger adverse reactions. This includes inflammation or allergies. Materials used are tested thoroughly. They undergo stringent regulations to ensure safety.
On the other hand, industrial-grade implants lack this level of scrutiny. These implants are made for general purposes, not for direct human contact. There might be unknown materials involved. The risk of cytotoxicity is higher with industrial-grade products. Even minor contaminants can pose serious health risks.
The choice between medical and industrial-grade implants requires careful consideration. Safety shouldn't be compromised. It’s essential to reflect on the long-term impacts. Adverse effects may not show up immediately. Understanding these differences can lead to better outcomes in orthopedic care. Choosing the right implant is a responsibility that demands attention.
This chart illustrates the key differences between medical grade and industrial grade orthopedic implants based on five critical factors: biocompatibility, safety testing, material purity, regulatory approval, and longevity. Medical grade implants generally score higher in these areas, reflecting their more stringent manufacturing and safety standards.
When it comes to orthopedic implants, durability and performance are paramount. Medical grade implants are designed to withstand the rigors of the human body. They endure stress, movement, and biological interactions. Industrial grade implants, while robust, may not offer the same level of biocompatibility. This can lead to complications in a surgical setting, affecting patient recovery.
A medical-grade implant often undergoes rigorous testing. This includes fatigue testing and corrosion resistance. Such thorough evaluations ensure longevity. In contrast, industrial-grade options may lack these stringent assessments. This can translate to riskier outcomes in surgeries. For example, a surgical implant that fails prematurely can lead to additional surgeries. This not only impacts healthcare costs but also patient well-being.
The choice between these two grades raises questions. Is the cost of medical-grade implants justified by their performance? Many argue that the higher price is worth the peace of mind. Yet, others suggest that in less demanding applications, industrial-grade options may suffice. This gray area in choice can lead to overthinking among practitioners. Balancing cost, performance, and patient safety can be a challenge.
: Medical-grade implants often use high-quality titanium or cobalt-chromium alloys. These materials ensure excellent biocompatibility and longevity.
Industrial-grade implants usually utilize more affordable materials like stainless steel or lower-grade titanium, which may lack necessary quality.
Regulatory standards help ensure safety and efficacy. Strict testing minimizes risks like allergic reactions and infections in patients.
Industrial-grade implants lack the rigorous testing of medical-grade ones, raising concerns about durability and potential safety issues.
Medical-grade implants are designed to endure stress and biological interactions. Industrial-grade may not offer the same level of biocompatibility.
They go through rigorous assessments, including fatigue testing and corrosion resistance, ensuring their longevity in surgical settings.
Many argue that the higher cost is justified by better performance and peace of mind, though some may prefer lower-cost options in specific cases.
A failed implant can lead to additional surgeries, which raises healthcare costs and affects patient recovery.
Yes, the type of implant can significantly impact recovery and overall well-being, making it an important decision for practitioners.
Yes, practitioners often overthink their choice, trying to balance cost, performance, and patient safety.
The article "Top 5 Differences Between Medical Grade and Industrial Grade Orthopedic Implants" delves into the crucial distinctions between these two categories of implants. What is the difference between medical-grade and industrial-grade orthopedic implants? Key characteristics of medical-grade implants include materials specifically designed for biocompatibility and safety, ensuring they can be safely used in the human body. In contrast, industrial-grade implants may not meet the same stringent regulatory standards or safety considerations.
Additionally, the materials used in these implants vary significantly, with medical-grade options often incorporating biocompatible metals and polymers, while industrial-grade may utilize lower-grade materials. Regulatory standards play a pivotal role in governing medical-grade implants, dictating manufacturing practices and safety protocols. Finally, durability and performance differences highlight that medical-grade implants are designed for optimal performance in surgical applications, ensuring longevity and patient safety. Understanding these distinctions is vital for healthcare professionals and patients alike when making informed decisions about orthopedic solutions.

 Daher Orthopedic Implants
Daher Orthopedic Implants