The global market for Anterior Cervical Plating Systems is witnessing an unprecedented surge, driven by an aging global population and the rising prevalence of degenerative disc diseases. As a specialized factory and exporter, we recognize that Anterior Cervical Discectomy and Fusion (ACDF) remains the gold standard for treating cervical spine instability. The commercial landscape is shifting from traditional rigid systems to dynamic and low-profile plating architectures that minimize soft tissue irritation and dysphagia.
In regions like North America and Europe, the demand is focused on high-precision titanium alloys and biocompatible coatings. Conversely, in emerging markets across Southeast Asia and Africa, there is a growing need for cost-effective, durable fixation systems that do not compromise on safety. As a leading manufacturer, we bridge this gap by offering medical-grade solutions that meet rigorous international benchmarks while maintaining competitive industrial pricing.

Our flagship Anterior Cervical Plate System is designed for maximum biomechanical stability. Featuring a slim profile and a secure locking mechanism, it reduces the risk of screw backing out—a common complication in spinal surgeries. The plate's surface is treated with specialized anodization to enhance osseointegration.
Modern surgeons require plating systems that support smaller incisions. Our R&D team focuses on narrowing plate widths and developing specialized instruments for MIS approaches to reduce patient recovery time.
The integration of AI in design allows for "Patient-Specific" plate contouring. Using 3D modeling, we can simulate stress distribution before the first titanium block is cut on our CNC machines.
The trend is moving towards combining rigid and semi-rigid fixation. Our latest systems allow for controlled settling, which promotes better load-sharing between the implant and the bone graft.
For international medical distributors and hospital procurement officers, selecting an Anterior Cervical Plating Systems Factory involves more than just price comparison. Key factors include:
Products must carry CE marking and ISO 13485 certification. As a trusted exporter, we provide a complete technical file and clinical evaluation reports for local registration.
Hospitals cannot afford surgical delays. Our factory maintains a robust inventory of standard sizes, ensuring rapid lead times for international shipments via air or sea.
Many partners require custom branding or modifications to instrument trays. Our facility is equipped to handle private label manufacturing with precision laser etching.
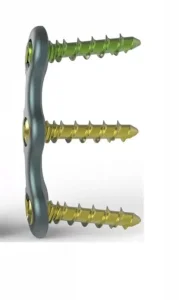
The Anterior Cervical Fixation System with Screw is optimized for multi-level fusion. Its integrated "One-Step" locking mechanism provides tactile feedback to the surgeon, ensuring the screw is fully seated and locked without the need for additional locking caps.
Choosing a Chinese factory for Anterior Cervical Plating Systems offers a unique combination of high-tech manufacturing and economic efficiency. In the past decade, China's orthopedic sector has undergone a massive transformation, moving from imitation to world-class innovation.
Raw Material Access: China is a global leader in titanium production. We have direct access to the highest grade medical titanium (Grade 23), ensuring our plates are both lightweight and incredibly strong.
CNC Precision: Our facility utilizes 5-axis German and Japanese CNC centers. This allows us to maintain tolerances within microns, crucial for the threading of cervical screws and the locking mechanisms of plates.
Global Logistics: Located near major shipping hubs, we offer seamless export services. Our documentation team is expert in handling the complexities of medical device customs clearance in Europe, South America, and the Middle East.
We are dedicated to the research, development, production, and sales of orthopedic implants and tools. In the continuous process of design updates, we strive for excellence, meticulously crafting each product.
Our quality control philosophy: Quality and integrity first, striving for excellence, and pursuing the highest standards.
A professional R&D team and long-term technical exchanges with leading hospital experts ensure that our products function to their fullest potential.
Our factory has passed ISO 9001 and ISO 13485 certifications. Our products exceed all international quality and safety standards, including CE and FDA certifications. For many years, our products have been exported to more than 70 countries and regions in Europe, America, Africa, and Southeast Asia.
Our professional team is dedicated to serving every customer. Your trust is the greatest recognition of our service. We will rely on high-quality products and continuously launch new marketable products based on market demand, achieving mutual benefit and win-win results with our customers.
